Willnow Lab
Molecular Cardiovascular Research
LRP2
iPSC-based modeling of human brain malformations
Introduction
Metabolic disorders not only impact health of the adult brain but also impair embryonic development of this tissue. This problem is exemplified by inheritable defects in cholesterol biosynthesis causing malformation of the forebrain (holoprosencephaly, HPE) in patients and animal models. In earlier work, we studied the significance of the low-density lipoprotein receptor-related protein 2 (LRP2), a presumed lipoprotein receptor in the developing forebrain and a genetic cause of human HPE. Contrary to prior hypotheses, we showed that LRP2 does not act as a receptor for cholesterol-rich lipoproteins in the CNS but as an auxiliary receptor for the cholesterol-modified morphogen sonic hedgehog (SHH) and that LRP2 deficiency results in loss of SHH signals required for forebrain patterning (Christ et al., Dev Cell 2012; Dev Cell 2016).
In recent years, we continued our investigations of LRP2 (dys)functions in gene-targeted mouse models. We documented loss of SHH signaling during heart development as the underlying reason for heart malformations in patients with inheritable LRP2 deficiency (Donnai-Barrow Syndrome/DBS; Christ et al., Hum Mol Genet 2020). We used single-cell RNA-Seq to uncover the importance of LRP2 for adult neurogenesis (Zywitza et al., Cell Reports 2018), and we collaborated with others to assess the importance of the receptor in fetal alcohol syndrome (Hong et al., eLife 2020) as well as muscle glucose metabolism (Seo et al., Nat Commun 2020).
iPSC-based modeling of human forebrain development uncovers mechanisms of HPE
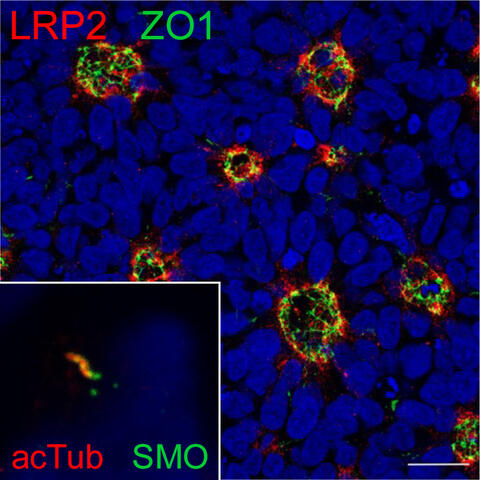
Fig. 7: Neural rosettes formed by iPSC-derived human neuroepithelial progenitors (NPCs). Cells are immuno-stained for apical markers LRP2 (red) and ZO1 (green). Nuclei are stained with DAPI (blue). Scale bar: 20µm. Inset shows a primary cilium, required for SHH signaling, immunostained for Smoothened (green) and acetylated tubulin (red) on NPCs
While transgenic mice have been indispensable for exploring the role of LRP2 in organogenesis, we also realized the limitations in recapitulating human brain development in rodents. Therefore, a major ambition of our recent work had been the development of iPSC-based models to study LRP2 functions in the human brain. Here, we applied iPSC-derived neuroepithelial progenitor cells (NPCs) that form neural rosettes. These recapitulate structural and functional aspects of the embryonic neural tube that develops into the various parts of the CNS, including the forebrain (Fig. 7). In our study by Flemming et al. (Kidney Int 2020), we used NPCs to document SHH-induced lysosomal decay of mutant LRP2 as cause of a novel familial form of DBS. In a second study, we uncovered the interaction of SHH and Notch signaling pathways in neural tube patterning, providing a molecular explanation for forebrain phenotypes shared by defects in both pathways (Marczenke et al., Development 2021).