Willnow Lab
Molecular Cardiovascular Research
SORLA
Linking amyloidogenic processes with insulin signaling
Introduction
SORLA is a 250 kDa receptor expressed in brain but also metabolic tissues, including visceral fat and pancreas. In earlier work, we focused on a role of the receptor in neurons, showing association of the encoding gene SORL1 with the common sporadic form of AD (Rogeava et al., Nat Genet 2007; Caglayan et al., Arch Neurol 2012). We identified the ability of SORLA to mediate lysosomal clearance of neurotoxic Aβ peptides, the etiologic agent in AD, as an activity underlying its protective function in the disease (Andersen et al., PNAS 2005; Dumanis et al., J Neurosci 2015). We developed mathematical models to assess the amyloidogenic burden associated with SORLA levels in neurons (Schmidt et al., EMBO J 2012), and we corroborated pharmacological induction of receptor expression as a therapeutic option in AD (Rohe et al., J Neurosci 2009). Perhaps most exciting, we documented loss of lysosomal Aβ sorting in a mutant SORL1 gene variant as novel cause of early onset AD, identifying it as the only known gene to cause both sporadic and familial forms of this disease (Caglayan et al., Sci Transl Med 2014).
SORLA is a clearance receptor to protect against islet amyloid deposition
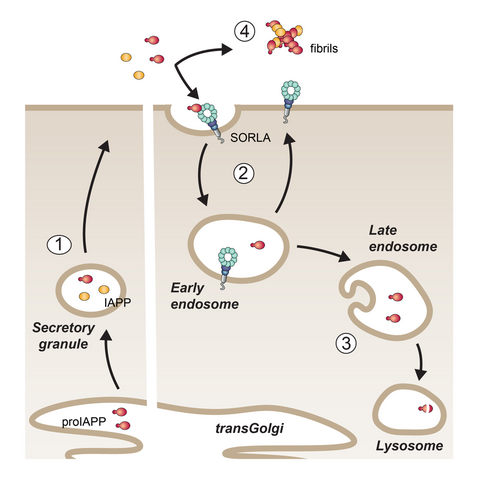
Fig. 2: In pancreatic islets, SORLA acts as clearance receptor for proIAPP co-secreted with IAPP (steps 1-3), reducing fibril deposits in T2D (step 4).
Type 2 diabetes (T2D) is a major risk factor for AD and both diseases share pathological hallmarks, including amyloid deposits that compromise cell viability and function. Amyloid deposits in the pancreas are composed of islet amyloid polypeptide (IAPP), a peptide hormone co-secreted with insulin. In case of insulin hypersecretion, as in T2D, excessive amounts of IAPP are released and deposited as fibrils that further compromise islet function. Given the expression of SORLA in islet β-cells, we reasoned that the receptor may have a similar protective function as in the brain, counteracting islet amyloid deposition. In line with our model, we showed that SORLA acts as clearance receptor for the pro-form of IAPP (proIAPP), co-released with the mature hormone (Fig. 2; step 1). SORLA directs re-uptake and lysosomal catabolism of proIAPP, but not IAPP (steps 2 and 3), counteracting amyloid deposition from proIAPP without compromising active hormone function (step 4) (Shih et al., Mol Metab 2022).
SORLA is an insulin-sensitizing receptor in control of obesity
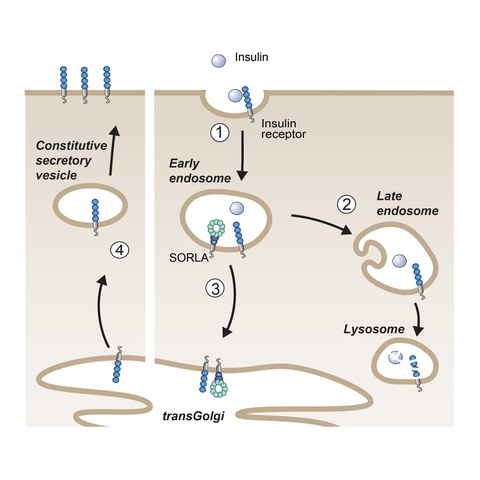
Fig. 3: SORLA shuttles internalized insulin receptors back to the cell surface (steps 1 and 3), bypassing lysosomal degradation (step 2) and sensitizing cells for insulin signaling (step 4).
As well as with AD, SORL1 also shows genome-wide association with the longitudinal risk of obesity, i.e., the risk for obesity in adults who have been overweight as juveniles. Given the dramatic prevalence of childhood obesity and the impact of consequential metabolic disturbances for risk of AD in adults, we investigated SORLA’s causal role in juvenile body weight control. Our studies were guided by our earlier work that SORLA acts as a sorting factor for the insulin receptor (IR) in adipose tissue (Schmidt et al., J Clin Invest 2016) (Fig. 3). In detail, insulin-bound IRs typically move from the cell surface to lysosomes to down-regulate hormone signaling. However, SORLA sorts internalized IRs via the TGN back to the cell surface, sensitizing cells for insulin signal reception. Using mouse models, we showed that SORLA is highly expressed in adipocyte progenitors in juvenile white adipose tissue, priming these cells for nutritional stimuli provided by insulin. Overfeeding causes SORLA-dependent expansion of this progenitor cell pool in juveniles, driving excessive fat cell production in adult life. The relevance of our findings was substantiated in human cohorts, in which SORLA levels correlated with visceral obesity (Schmidt et al., J Cell Biol 2021).