Sommer Lab
Intracellular Proteolysis
Ubiquitin conjugation in muscle atrophy
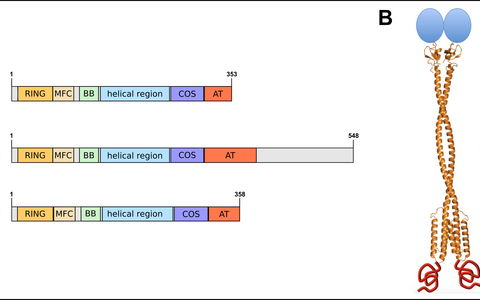
Many conditions such as cachexia, malnutrition and inactivity e.g. because of prolonged bedrest after surgery lead to enhanced degradation of sarcomeric proteins resulting in the reduction of muscle mass, the so-called muscle atrophy. Crucial mediators of protein turnover of the sarcomere and thus of muscle atrophy are the three muscle-specific RING finger proteins MuRF1 – 3 (Figure 1A & B).
Figure 1: MuRF domains and topology. (A) The three MuRF proteins share a common domain organization comprised of the two Zn2+ binding RING and B box-type domains followed by a coiled coil helical region and an acidic tail. Furthermore, a so called MFC domain is conserved between all three MuRF proteins (BB: B box-type domain, MFC: MuRF family conserved domain, AT: acidic tail; adapted from Centner et al., 2001 & Witt et al., 2005). (B) MuRF proteins form homo- or heterodimers of approximately 10 nm in length (Model of the MuRF1 homodimer taken from Franke et al., 2014).
MuRF1 protein levels increase upon atrophy induction. MuRF1 has been implicated in the turnover of sarcomeric proteins like titin and Myosin heavy chain. It was therefore anticipated that MuRF1, and possibly also MuRF2 and 3, act as autonomous RING-type ubiquitin ligases, which directly bind and ubiquitylate their client proteins. By using C2C12 cells as a model to study sarcomeric protein quality control, muscle atrophy and the role of MuRF proteins within these processes, we now show that MuRF1 constitutes a subunit of a large cullin-type ubiquitin ligase. Cullin-type ubiquitin ligases are modular protein complexes that contain combinations of substrate recruiting and substrate processing factors. These factors assemble on a so-called cullin scaffold protein. Notably, MuRF1 in complex with a previously undescribed partner protein DCAF8 appears to assist in substrate recruitment rather than to directly participate in the ubiquitylation reaction. This finding is of particular interest for the development of therapeutic strategies to encounter disease-related muscle atrophy.
We are continuing our cell culture-based work to characterize the formation of the MuRF1-cullin complex, its regulation and its substrate specificity in more detail. We are also currently establishing a powerful cell-free system to investigate the complex machinery that facilitates MuRF1-dependent protein ubiquitylation in vitro. Furthermore, we are interested to determine the contribution of MuRF2 and MuRF3 in muscle proteostasis and atrophy.