Poor delivery service in the brain
What causes Alzheimer’s disease is still unclear. We know that nerve cells in the brain die off in large numbers. We also know that plaque deposits, insoluble clumps of a protein called beta-amyloid, collect between the cells. In addition, it is known that tau fibrils are found inside the cells. In Alzheimer’s patients, tau is chemically altered such that protein fibers develop that result in cell death.
Nothing can keep neurons from dying
Finally, we are also aware of a genetic cause for increased risk of age-related dementia: people with a certain variety of the apolipoprotein E4, called ApoE4, are at much greater risk than those who have either ApoE3 or the rarer ApoE2. ApoE is a kind of delivery service for the brain. It supplies neurons with nutrients like fatty acids, which they need for proper synaptic function.
Despite all this knowledge, we still do not understand how Alzheimer’s begins. Nor are we anywhere close to developing effective treatments, even though all three of the proteins – beta-amyloid, tau, and ApoE – have been the target of new, very promising therapeutic agents. So far, nothing has been able to keep neurons from dying.
Not enough fat for the synapses
When nerve cells receive too few lipids, it disrupts the functioning of their synapses,” the biophysicist explains.
A new approach is now being taken by Dr. Melissa Birol, head of the Single-Molecule Biophysics Probing Quantitative Neuroscience Lab at the Berlin Institute for Medical Systems Biology of the Max Delbrück Center (MDC-BIMSB). Experts consider it so promising that the American-based Alzheimer’s Association has given Birol a total of about 150,000 U.S. dollars of funding over three years for her research endeavor. The Alzheimer’s Association is the largest non-profit health organization in the world devoted to studying Alzheimer’s and providing care and support for those who suffer from it.
“We suspect that tau and ApoE4 join together for some reason, interrupting the transport of fat both between the astrocytes and the neurons and within the nerve cells themselves,” Birol says. Astrocytes are star-shaped cells that surround neurons, providing them with structural and metabolic support. “When nerve cells receive too few lipids, it disrupts the functioning of their synapses,” the biophysicist explains. “As a result, the neurons can no longer communicate with one another, triggering a cascade of cellular events that end in neuron death.”
Disrupted transport to the cells
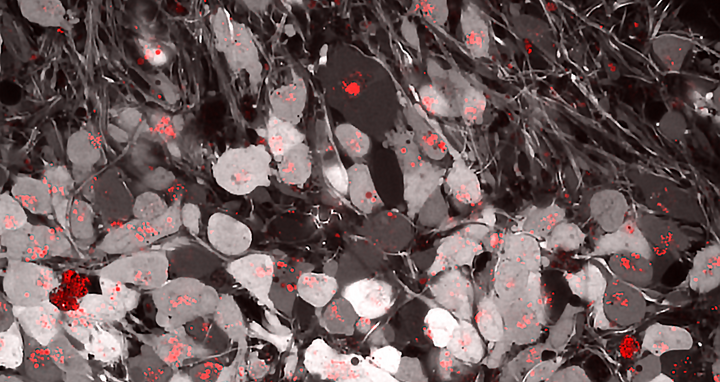
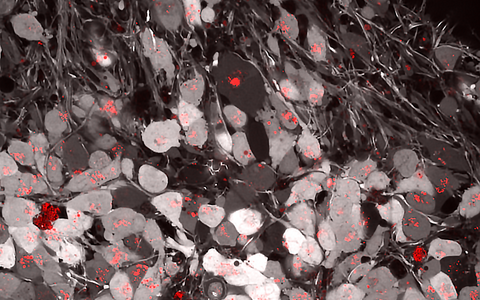
Birol and her team discovered this possibly very fatal interaction between tau and ApoE while performing experiments with brain organoids, miniature organs grown in the lab. “We observed that the two proteins already agglomerated outside the nerve cells,” reports Birol. “Inside the cells, we found them together in minuscule droplets of fat that – depending on the ApoE variant – spread in the cells at varying speeds and following different patterns.” Fat droplets with ApoE4 and tau were initially largely mobile inside the cells, unlike those with ApoE3. Over time, the dynamics of these droplets decreases, potentially ages, and correlates with the build-up of lipid storage units, known as lipid droplets.
Accumulation of Alzheimer’s associated protein, Tau, in a human brain organoid: The organoid was labeled grey reporting on the health of neurons and red for tau accumulations in the cell bodies.
“Our goal is now to understand the biophysical processes that allow these masses of ApoE and tau proteins to coalesce in individual cells and in the organoids,” Birol says. To do so, the research team plans to apply a combination of state-of-the-art methods of single-molecule spectroscopy and electron microscopy. In addition, they will study the lipid profile of neurons. Using functional calcium imaging, they will examine the extent to which the interaction of ApoE4 and tau in lipid droplets changes fat metabolism in brain cells, disrupts fat transport, and thus impairs the functioning of nerve cells and their synapses.
New approaches for more effective therapies
“By improving our understanding of the molecular basis for the development of Alzheimer’s disease, we hope to shed light on new therapeutic strategies that focus on the toxic agglomeration of lipids and proteins,” Birol says. Perhaps then the mass death of neurons can finally be stopped.
Text: Anke Brodmerkel