Neuroscience Clinical Research Center
Charité Campus Mitte
Tel: +49 (0)30 450 539 705
Fax: +49 (0)30 450 539 915
friedemann.paul@charite.de
Experimental and Clinical Research Center
Lindenberger Weg 80
13125 Berlin
MS is causally based upon autoimmune processes; this means that the body’s own immune system mistakes the nervous system as “foreign” and initiates an inflammation reaction, as a result of which nerve cells and axons, with their sheathing protective coating, are damaged. Many different neurological symptoms can occur. People suffering from MS can for example feel tingling sensations or numbness, receive paralyses or balance disorders, stumble more frequently or develop problems with their sight.
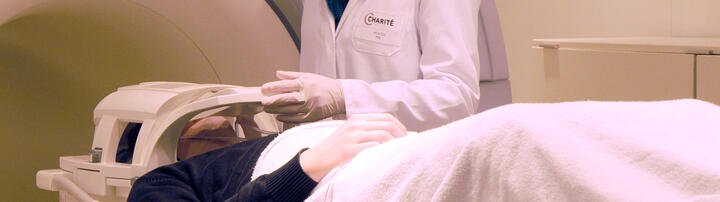
The course taken by MS varies greatly between individuals, and its course cannot be predicted well in individual patients. One of our fields of activity is the development and establishing of new progress and prognosis markers. These include, for example, the improvement of possibilities in the field of magnetic resonance imaging (MRI), e.g. ultra-high-field MRI and also the establishing of new imaging procedures such as optical coherence tomography (OCT).
It is not yet possible to cure MS. Progression and symptoms can, however, be positively influenced by various drug therapies and non-drug measures. Another fields of activity is the development and conducting of clinical studies with the aim of improving the therapeutic possibilities of MS. These include on the one hand clinical studies of phases I to IV, which we partly carry out in cooperation with the industry, and on the other hand new non-drug treatment approaches such as repetitive transcranial magnetic stimulation for the treatment of depressiveness and fatigue and visual restitution therapy for the improvement of persisting sight impairments after inflammation of the optic nerve.
Other diseases on which we have a research focus are Susac syndrome and neuromyelitis optica.
The Clinical Neuroimmunology group works in close cooperation with the interdisciplinary outpatient clinics of the Experimental and Clinical Research Center (ECRC). In addition to the Neuroimmunology outpatient clinic, other additional disciplines ensure state-of-the-art patient care based on the current state of knowledge.
For more information, explore MDC’s latest findings in the MS field.
Prof. Dr. med. Friedemann Paul, Facharzt für Neurologie
friedemann.paul@charite.de
PD Dr. Tanja Schmitz-Hübsch, Leader Research Outpatient Clinic
tanja.schmitz-huebsch@charite.de
studienambulanz-ag-paul@charite.de
Dr. med. Judith Bellmann-Strobl, Leader Neuroimmunological Outpatient Clinic
judith.bellmann-strobl@charite.de
hochschulambulanz-buch@charite.de
PD Dr. rer. nat. Chotima Böttcher, Leader Laboratory Research
Chotima.boettcher@charite.de / ORCID
Jana Bigall, Studynurse Team Lead
jana.bigall@charite.de
Vanessa Tatum, Project Management Lead
vanessa.tatum@charite.de
PD Dr. rer. nat. Chotima Böttcher, Team Leader: Single-cell and Spatial Profiling in Neuroimmunology. Chotima.boettcher@charite.de / ORCID
Adeline Jeanne Dehlinger, M.Sc., PhD student adeline.dehlinger@charite.de
Dr. Camila Fernández Zapata, Postdoctoral fellow maria.fernandez@charite.de / ORCID
Christian Böttcher, Biological Technician, Biomaterials and protein analysis Christian2.boettcher@charite.de
Gerardina Gallaccio, M.Sc., PhD student gerardina.gallaccio@charite.de
Meng Wang, MD., PhD student meng.wang@charite.de
Wenbo Sun, MD, PhD student wenbo.sun@charite.de
Anna Holovach, MD student anna.holovach@charite.de
GueHo Jang, M.Sc., PhD student gueho.jang@charite.de
Wenbo Zhang MD student, wenbo.zhang@charite.de
Lisa- Marie Diekmann PhD student, lisa-marie.diekmann@charite.de
Juniorprof. Dr. Hanna Gwendolyn Zimmermann, Team Leader: Visual System Research
hanna.zimmermann@charite.de
Lukas Reeß, Optometrist
Carla Leutloff, Optometrist
Gilberto Solorza Buenrostro, Physician
Prof. Dr. med. Friedemann Paul, Team Leader: MRI Team
friedemann.paul@charite.de
Dr. Claudia Paarmann-Chien, Senior Postdoc
claudia.paarmann-chien@charite.de
Dr. Lina Anderhalten, Postdoctoral fellow
Darius Mewes, MD/PhD Student
Henri Trang, PhD Student
Qianlan Chen, PhD Student
Michael Sheel, Neuroradiologist
Susan Pikol, Medical Technical Assistant
Cynthia Kraut, Medical Technical Assistant for Radiology
Dr. Maria Hastermann — Team Leader: Adaptive Immunity and Novel Therapies. Maria.hastermann@charite.de / ORCID
Our Team
Sara Cestari, M.Sc., PhD Student
Ekaterina Vasileva, M.Sc., PhD Student
ekaterina.vasileva@charite.de
Dr. Nadine Strempel — Senior Postdoc
Melina Quast, MD/PhD Student
We are looking for new people! Write us an email with your intended position, CV and motivation letter.
Jana Bigall, Studynurse Team Lead
jana.bigall@charite.de
studienambulanz-ag-paul@charite.de
Mirjana Mitrovic, Office — Research Outpatient Clinic
Katharina Stößlein, Studynurse — Research Outpatient Clinic
Claudia Messelhäußer, Studynurse — Research Outpatient Clinic
Luisa Kegel, Studynurse — Research Outpatient Clinic
Bibiane Seeger-Schwinge, Medical Technical Assistant — Laboratory
Nora Seelig, Medical Technical Assistant — Laboratory
Dr. Lina Samira Bahr, Team Lead Metabolic Team
lina.bahr@charite.de
Dr. Pia Sperber, Team Lead Data Management
pia.sperber@charite.de
Martin Weygandt, Scientist
Susanna Asseyer, Physician
Lil Meyer-Arndt, Physician
Dr. med. Judith Bellmann-Strobl, Leader Neuroimmunological Outpatient Clinic
judith.bellmann-strobl@charite.de
hochschulambulanz-buch@charite.de
Dr. Uta Hoppmann, Neurologist
Dr. Janina Behrens, Neurologist
Philippe Manceau, Neurologist
Gritt Stoffels, Studynurse
Dr. Joseph Kuchling, Physician
Anja Mähler, Leader Nutritional Consultation
Dr. Nadja Siebert, Neurologist
Elena Steinle, Studynurse
Neuroinflammation is a common feature of autoimmune diseases of the central nervous system (CNS) such as multiple sclerosis (MS), neuromyelitis optica spectrum disorders (NMOSD) and myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD), in which the pathologies are associated with various immune responses in different body compartments e.g. local inflammation, microglial activation, and CNS or gut infiltration of circulating immune cells. Characterization of more diverse immune cell types residing in different body compartments including gut, peripheral blood, cerebrospinal fluid (CSF), brain interface and brain parenchyma is required for better understanding dynamic compartmentalization of these cells in early as well as in the progressive stages of diseases.
In our team, we characterize the metabolomic and proteomic profiles of body fluid (i.e. plasma and CSF) that associate with functional and phenotypic changes of immune cells in gut, peripheral blood, CSF and the CNS of MS, NMOSD and MOGAD patients/donors, using mass cytometry and imaging mass cytometry. We aim to assess dynamic cell signalling of immune cells from different compartments using a combination of deep protein and metabolomics profiling with phenotypic profiling, in order to unravel neuroinflammation-associated phenotypic transmission of immune cells and how these cells are involved in CNS homeostasis.
Diagnosis, differential diagnosis, monitoring of disease course and treatment effects in patients with autoimmune inflammatory CNS diseases such as multiple sclerosis (MS), neuromyelitis optica spectrum disorders (NMOSD), or the newly emerging disease entity myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD) still pose significant challenges for neurologists and evoke anxiety in affected patients. Our work focuses on the contribution of conventional and advanced structural and functional MRI techniques (as well as visual system research, immune cell profiling and serum derived biomarkers (for example, aquaporin‑4 (AQP4) antibodies, myelin oligodendrocyte glycoprotein (MOG) antibodies) to diagnosis and differential diagnosis of autoimmune CNS diseases. Moreover, we use these methods to deepen our understanding of the association of structural and functional CNS changes with clinical symptoms (using state of the art and innovative measures of neurological disability, cognitive dysfunction, visual function), as well as with disease course and treatment response. Imaging (and biomarker) studies are embedded into ongoing longitudinal observational (i.e. BERLimmun (including CIS, MS, NMOSD and MOGAD patients), acute optic neuritis (ACON)) studies with >500 recruited patients, yielding clinically and paraclinically extremely well phenotyped cohorts, and are applied in interventional trials (neuroprotective, symptomatic) with the aim of broadening treatment options.
Using quantitative multi-parameter mapping (MPM), MRI studies can investigate clinically relevant microstructural changes with high reliability over time and across subjects and sites. We have established a fast MPM protocol with 3D multi-echo fast low angle shot (FLASH) acquisitions including post-processing optimized for longitudinal clinical studies to obtain after reconstruction the following whole-brain MPM maps with 1.6 mm isotropic resolution: proton density (PD), magnetization transfer saturation (MT), longitudinal relaxation rate (R1), and transverse relaxation rate (R2*). The intra-subject stability of the 1.6‑mm MPM protocol is 2 – 3 times higher than for the standard 1‑mm sequence and can be achieved in less than half the scan duration. By combining Gibb’s ringing correction and bias field correction using acquired B1+ maps we achieved excellent stability of a 7‑minute MPM sequence. The MPM sequence and a corresponding multi-shell diffusion-weighted imaging (DWI) sequence derived from the human connectome project and suitable to calculate Neurite Orientation Dispersion and Density Imaging (NODDI) are part of the core imaging protocol of the ongoing Berlimmun study.
We are working on this with a translational approach; this means that we try to transfer new developments and findings from basic research directly into clinical work.
The Clinical Neuroimmunology Group is a part of the Experimental and Clinical Research Center (ECRC), a cooperation between the MDC and the Charité — Universitätsmedizin Berlin. For patients interested in our group, please refer to our Neuroimmunology outpatient clinic.
Berlin Registry of Neuroimmunological Entities (BERLimmun) — a monocentric prospective observational study on the long-term course of neuroimmunological diseases.
Sponsor: IIT
Recruitment: yes
Local PI: PD Dr. Tanja Schmitz-Hübsch
Study Number: German Clinical Trial Register DRKS00026761
Acute Optic Neuritis Study (AON‑S), a monocentric, prospective observational study on symptomatology, diagnosis, therapy and prognosis of acute optic neuritis.
Sponsor: IIT
Recruitment: yes
Local PI: Dr. Susanna Asseyer/Prof. Dr. Friedemann Paul
Study Number: NCT05605951
Neural mechanisms of mental and emotional processes in multiple sclerosis and their relevance to disease severity.
Sponsor: IIT
Recruitment: yes
Local PI: PD Dr. Martin Weygandt
A Phase 3, randomized, double-blind, efficacy and safety study comparing SAR442168 to placebo in participants with nonrelapsing secondary progressive multiple sclerosis (HERCULES).
Sponsor: Sanofi
Recruitment: Screening closure 29.11.2022
Local PI: Prof. Dr. Friedemann Paul
Study Number: EudraCT 2020−000647−30
A Phase 3, randomized, double-blind, efficacy and safety study comparing SAR442168 to placebo in participants with primary progressive multiple sclerosis (PERSEUS).
Sponsor: Sanofi
Recruitment: yes
Local PI: Prof. Dr. Friedemann Paul
Study Number: EudraCT 2020−000645−14
Sponsor: IIT
Recruitment: yes
Local PI: Prof. Dr. Stefan Gold
Clinical validity of confirmed disease progression defined by marker-less home-based assessment of motor function in multiple sclerosis (HOPE-MS).
Sponsor: IIT (Celgene)
Recruitment: yes
Local PI: PD. Dr. Tanja Schmitz-Hübsch
Tracking the immune response to SARS-CoV‑2 modRNA vaccines in an open-label multicenter study in participants with relapsing multiple sclerosis treated with ofatumumab s.c. (KYRIOS).
Sponsor: Novartis
Recruitment: closed
Local PI: Prof. Dr. Friedemann Paul
A phase III, randomized, double-blind, placebo-controlled, multicenter study to evaluate the efficacy, safety, pharmacokinetics, and pharmacodynamics of satralizumab as monotherapy or in addition to baseline therapy in patients with myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD).
Sponsor: Roche
Recruitment: yes
Local PI: PD. Dr. Tanja Schmitz-Hübsch
Study Number: NCT05271409
A Randomized, double-blind, Placebo-controlled, multicenter Phase 3 pivotal study to evaluate the efficacy and safety of rozanolixizumab to treat adult patients with Myelin Oligodendrocyte Glycoprotein (MOG) Antibody-Associated Disease (MOG-AD).
Sponsor: UCB
Recruitment: yes
Local PI: Prof. Dr. Friedemann Paul
Study number: NCT05063162
Sensitivity of quantitative motor performance measures in multiple sclerosis – a prospective cohort study over two years using Microsoft Kinect.
Sponsor: IIT
Recruitment: closed
Local PI: Prof. Dr. Friedemann Paul
7 Tesla MRI in patients with multiple sclerosis and other neuroimmunological disorders.
Sponsor: IIT
Local PI: Prof. Dr. Friedemann Paul
Prevention of paclitaxel-related neurologic adverse events with lithium — a randomized, double-blind, placebo-controlled, exploratory proof-of-concept phase II clinical trial for the prevention of chemotherapy-induced neurotoxicity.
Sponsor: IIT
Recruitment: yes
Local PI: Prof. Dr. Matthias Endres
MR elastography in patients with multiple sclerosis.
Sponsor: IIT
Local PI: Prof. Dr. Ingolf Sack
Sponsor: IIT
Recruitment: yes
Local PI: Prof. Dr. Stefan Gold/Prof. Dr. Isabel Dziobek
Database on family planning and pregnancies in multiple sclerosis and Neuromyelitis optica spectrum disease (PreCoMS).
Sponsor: IIT
Local PI: Prof. Dr. Friedemann Paul
Specified laboratory investigations in Portugal.
Sponsor: IIT
Recruitment: yes
Local PI: Prof. Dr. Friedemann Paul
Regulatory T cells in Neuromyelitis optica.
Sponsor: IIT
Recruitment: yes
Local PI: Dr. Maria Hastermann
Determining the accuracy, reliability, validity, and responsiveness of video-operceptive motion analysis with Microsoft Kinect — VALKINECT.
Sponsor: IIT
Recruitment: yes
Local PI: Prof. Dr. Friedemann Paul
Care Model for Patients with Chronic Fatigue Syndrome (ME/CFS).
Sponsor: IIT
Recruitment: yes
Local PI: Prof. Dr. Carmen Scheibenbogen
Patient registry of the German Multiple Sclerosis Society, Federal Association (DMSG) within the MS Research and Project Development GmbH (MSFP), DMSG MS-Register.
Sponsor: DMSG
Recruitment: yes
Local PI: Prof. Dr. Friedemann Paul
Family planning and pregnancies in multiple sclerosis (PreCoMS)
Sponsor: IIT
Recruitment: yes
Local PI: Dr. Nadja Siebert
Study Number: NCT05010902
Analysis of the Pathophysiology and Pathology of Corona Virus Disease 2019 (Pa-COVID-19).
Sponsor: IIT
Recruitment: yes
Local PI: Prof. Carmen Scheibenbogen / Dr. Judith Bellmann-Strobl
Dynamic vascular analysis of the retina in the Pa-COVID-19 study.
Sponsor: IIT
Recruitment: yes
Local PI: Prof. Dr. Friedemann Paul
A study to investigate improvement in physical function in SF-36 with Vericiguat compared with Placebo in participants aged 18 to 50 years with post-COVID-19 syndrome (PCS and PCS/CFS).
Sponsor: IIT (Bayer)
Recruitment: yes
Local PI: Dr. Judith Bellmann-Strobl
Study Number: EudraCT 2021−005494−11 NCT05697640