Methods
Electrophysiology
Five electrophysiology rigs are set up in the laboratory.
Two setups are devoted primarily to extracellular recording from sensory afferents innervating the skin in vitro. This preparation was originally developed to record from nociceptive afferent fibers in the rat (Reeh, 1986). Martin Koltzenburg and myself (Gary Lewin) developed this preparation for recording from mouse and embryonic chick afferent neurons innervating skin (Airaksinen et al. 1996; Koltzenburg et al. 1997; Koltzenburg and Lewin, 1997). This preparation has the advantage that one can quantitatively measure changes in the physiological properties of functionally defined afferent neurons in animals subjected to a targeted mutation of any given gene.
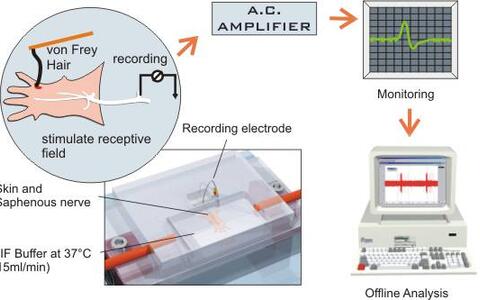
Fig1. A schematic diagram of the skin nerve preparation
Fig1. A schematic diagram of the skin nerve preparation (drawing by Paul Heppenstall)
We also currently have two electrophysiological set-ups devoted to whole cell patch clamp recording from acutely-isolated mouse dorsal root ganglion neurons (A new set-up is currently being built). Using this preparation, one can monitor biophysical properties, such as voltage activated currents, and action potential parameters of neurons taken from mice with targeted disruption of specific genes (see research)
Molecular Biology
A variety of standard molecular biological techniques are used in the laboratory. Molecular cloning is used to isolate DNA sequences as templates to make cRNA probes for northern blotting and in situ hybridization experiments. We have used 5′ RACE techniques to isolate novel cDNA sequences. In order to directly assess the function of genes in specific biological processes (such as sensory mechanotransduction) we have also generated knockout mouse models.
Cell Biology
We routinely use cultured cell lines and primary sensory neurons from chicken, mouse and rat. We have also established cell lines that overexpress epitope tagged versions of mammalian mec gene homologues. We use such methods to express ion channel proteins together with proteins that might modulate the kinetic properties of these channels. Cell lines are examined using patch clamp techniques