Projects
Research Projects
The last time you smelled a cookie and saliva was released into your mouth TMEM16A, a calcium activated chloride channel (CaCC), which was discovered recently, was involved. Its sequence is not related to any other ion channel or transporter and it is the founding member of a new protein family.
Calcium activated chloride currents have first been described in salamander photoreceptors in 1982, but the molecular identity remained elucive until last year. CaCCs are not only important for membrane potential stabilization in photoreceptors of the retina and the secretion of fluids in glands and airway epithelia. They have also been implicated in a wide range of other physiological functions including the high-gain, low-noise amplification in olfactory transduction, taste adaptation, control of action potential waveform in neurons and positive feedback regulation of smooth muscle contraction induced by G‑protein coupled receptors that is important to control vascular tone or uterus contraction.
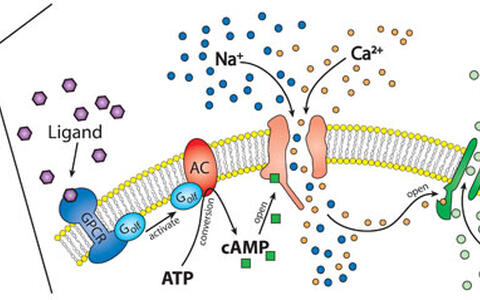
One of the best understood physiological functions of CaCCs is their role during the smelling process in in ciliar membranes of olfactory receptor neurons as depicted in the following figure:
Binding of odorants to specific G‑protein coupled receptors activates an adenylate cyclase (AC) through the G‑protein α‑subunit Golf. The produced cAMP binds to and opens cyclic nucleotide gated channels and calcium enters the cell. The initial depolarization caused by the opening of this unselective cation channels is than amplified by the opening of CaCCs. It has been estimated that the chloride current can be up to 30 times larger than the cation current.
An other example for CaCC function is the role in some vascular and airway smooth muscle cells where they appear to be involved in contraction stimulated by agonists and possibly blood pressure regulation. Ligand binding to G‑protein coupled receptors causes the release of calcium from the sarcoplasmatic reticulum through the phospholipase C pathway. Calmoduline activates myosin light chain kinase after Ca2+ binding and causes the muscle to contract. However in a positive feedback loop Ca2+ also activates CaCCs and the Cl- efflux causes the cell to depolarize and voltage gated calcium channels to open. The additional calcium influx is responsible for up to 30% of the contraction and hence the use of blockers of CaCC in such smooth muscle cells might benefit patients suffering from hypertension, asthma or bronchitis.
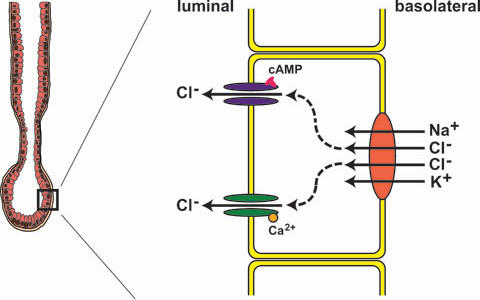
The role of CaCCs in epithelial cells is very different. Here they play a role in fluid secretion. In a simplified model chloride is taken up by the Na+/K+/2Cl- cotransporter at the basolateral side of epithelial cells in airways or exocrine glands. After passing the cell it enters the lumen through chloride channels. Water will than follow passively.
Most epithelial cells express two types of chloride channels: The cAMP regulated cystic fibrosis transmembrane conductance regulator (CFTR) and CaCC. In many exocrine glands, e.g. salivary glands, and also in mouse airway epithelia CaCCs dominate the chloride conductance. Because of this it has been suggested to use openers of CaCCs to bypass the defect CFTR channels in cystic fibrosis patients.
Though a lot is known about CaCCs, the physiological function in many tissues, e.g. in the retina, remains sketchy. One reason for limited progress in the field of CaCCs was the leak of knowledge about the molecular identity of CaCCs. Over the last 25 years several candidates have been proposed to be the molecular correlates of CaCCs. However none of them shared the hallmark features or expression profile of most native currents. This includes a strong rectification at submaximal Ca2+ concentrations, which disappears under saturating conditions. Using a new expression cloning strategy involving Axolotl oocytes, TMEM16A and TMEM16B have recently been shown to be CaCCs. They are the founding members of a new family of ion channels with no homology to any other known ion channel or transporter. TMEM16 proteins are expressed in a wide range of tissues ranging from neurons over smooth muscles to connective tissue and epithelia. Research in our laboratory focuses on the TMEM16 family of transmembrane proteins.
The TMEM16 family has 10 members in mammals, 5 in fly and 1 in yeast. Beside the function of TMEM16A as CaCC little is known about this family. Mice containing a deletion in the TMEM16A gene die few weeks after birth and suffer from tracheomalacia, a development phenotype of the lung and Gnathodiaphyseal dysplasia, a rare disease characterized by bone fragility, sclerosis of tubular bones and lesions of the jawbone, is caused by mutations in the human TMEM16E gene. But the role of TMEM16E in healthy bone and how its disruption leads to the phenotype remain elusive. TMEM16J shows a very strong regulation by the tumor suppressor p53 and some TMEM16 proteins are highly amplified in specific forms of cancer.
Given the general role of chloride channels in mammalian physiology and the possibility that all TMEM16 proteins are involved in ion transport, we are interested in
— the biophysical functions / properties of TMEM16 proteins,
— the regulation and interaction partners of this proteins,
— the physiological function, in particular for sensory transduction, smooth muscle contraction and fluid secretion in epithelia,
— potential involvement in human inherited diseases.
Suggested readings:
Schroeder BC, Cheng T, Jan Y, Jan LY, (2008). Expression cloning of TMEM16A as a calcium activated chloride channel subunit. Cell, 134, 1019 – 1029.
Hartzell HC, Yu k, Xiao Q, Chien LT, Qu Z, (2008). Anoctamin / TMEM16 family members are Ca2+ activated Cl- channels. J Physiol [Epub ahead of print] (review)
Hartzell C, Putzier I, Arreola J (2005), Calcium activated chloride channels. Annu Rev Physiol, 67, 719 – 758. (review).