An emergency call to blood stem cells
During infections, inflammations, and other kinds of stress, the body needs to produce large quantities of specific kinds of blood cells very quickly. This task is accomplished by haematopoietic stem cells (HSCs) that can generate all the cell types that make up blood. Researchers know that proteins called transcription factors are a major factor in determining the fate of the offspring of HSCs: These molecules change cells’ patterns of active and silent genes and lead them to specialize in particular ways. Molecules called cytokines, which are secreted by cells during times of stress, also contribute to blood cell development. Their main role, most scientists have believed, is to stimulate the survival and replication of blood cells that have already specialized. Now Michael Sieweke’s lab at the MDC and the Center of Immunology Marseille-Luminy has found evidence that cytokines directly influence the way HSCs produce specialized offspring by activating key transcription factors. The work, published in the May 9 issue of Nature, changes the textbook view of how HSCs respond to inflammations and other challenges and has important implications for therapies involving stem cells.
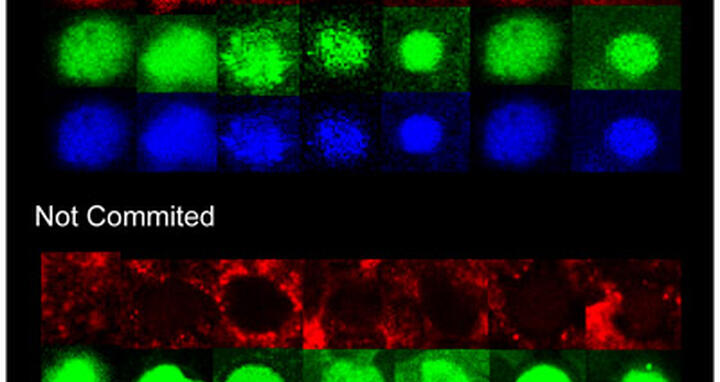
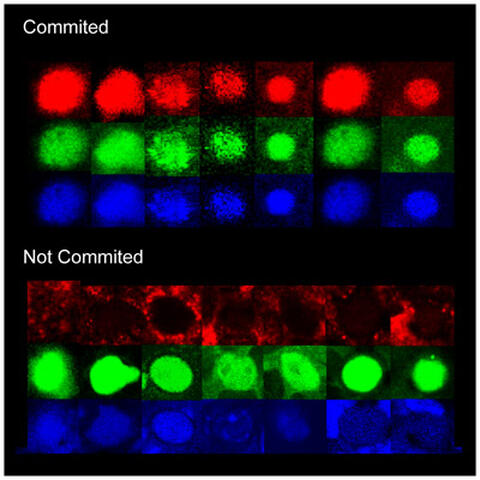
A Blood Stem Cell “ID Card:” each blood stem cell (HSC) is identified in vivo by its nucleus (in blue), the total protein content in cytoplasm (green) and the expression level of PU.1 (red). HSCs that lack M‑CSF stay uncommitted (as seen by the absence of PU.1, in the lower panel). In the presence of M‑CSF (upper panel) HSC express massive amounts of PU.1, leading to the production of the types of white blood cells needed to fight an infection.
Noushine Mossadegh-Keller and Sandrine Sarrazin have been studying a transcription factor called PU.1. This protein is a “master regulator” that triggers HSCs to develop into myeloid types such as macrophages, the large white blood cells that digest bacteria and other foreign particles.
“PU.1 and other master regulators are powerful ‘switches’ that send HSCs along particular developmental routes,” Michael says. “It hasn’t been clear what initially activates these switches – whether some sort of external signal is involved, or whether it happens through random internal variations.”
There were some hints that cytokines might play a role. When tissues are infected or experience physiological stress, they secrete large quantities of cytokines. This process is accompanied by a strong increase in the production of mature blood cells. Another hint came from HSCs that lacked an important transcription factor called MAFB. An earlier study by Michael’s lab had shown that HSCs without this molecule became more sensitive to cytokines. When such cells are exposed to a cytokine called M‑CSF, the master regulator PU.1 becomes active.
This suggested that M‑CSF and PU.1 were linked together in a signaling network that controlled the development of stem cells and that the cytokine might be throwing the master switch.
To determine whether this was the case, the scientists used HSCs from mice with a fluorescent tag added to the PU.1 gene. Now when the cells produced this protein, the molecule would glow and become visible under the microscope. Then they exposed HSCs to M‑CSF. When the HSCs detected the cytokine, the number of cells that had switched on PU.1 rose significantly. These cells also turned on other myeloid-specific genes and they began differentiating into myeloid cells. Further experiments demonstrated that the cytokine was actually changing the cells’ identity, rather than amplifying or enhancing survival of cells that had already started along the myeloid developmental pathway. .
Previous experiments had taken place in cell cultures containing huge numbers of cells; in that context, the fates of cells might be due to complex interactions in a community of cells. Noushine and Sandrine wanted to be sure that they weren’t observing some sort of “population effect.” To rule this out, they carried out their experiments on single HSCs using several new methods. These techniques allowed the scientists to conclude that M‑CSF was indeed activating PU.1, the master switch for producing myeloid cells.
“This shows that under stress conditions of infection, high levels of M‑CSF directly stimulate the development of myeloid cells from HSCs,” Michael says. “This challenges the current view both of the functions of cytokines in this process and the way the stem cells make differentiation decisions. In the past we’ve noted that HSCs divide more rapidly in response to bacterial or viral infections; our results now suggest that certain types of infections may also prompt the tailored development of specific types of cells needed to respond to the challenge.”
- Russ Hodge
Note: The Sieweke group is based at the Center of Immunology Marseille-Luminy in southern France and joined the MDC as part of the Helmholtz-INSERM Franco-German cooperation initiative. The group’s homepage at the link below provides an excellent introduction to haematopoietic stem cells and their differentiation for non-specialists.
Highlight Reference:
Mossadegh-Keller N, Sarrazin S, Kandalla PK, Espinosa L, Stanley ER, Nutt SL, Moore J, Sieweke MH. M‑CSF instructs myeloid lineage fate in single haematopoietic stem cells. Nature. 2013 May 9;497(7448):239 – 43.