Leutz Lab
Cell Differentiation and Tumorigenesis (Emeritus)
Profile
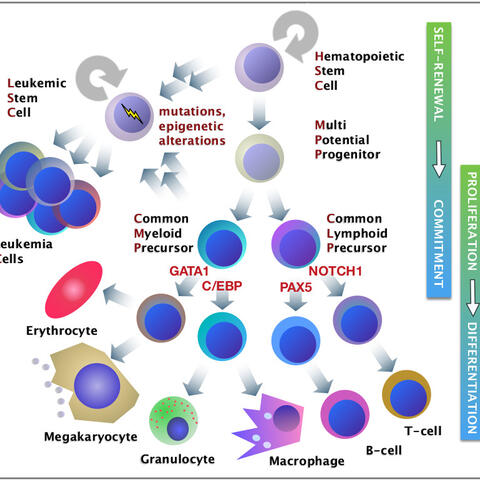
The inherent self-renewal property of hematopoietic stem cells vanishes during differentiation but may reoccur in tumor cells (see “Research” for details;). Key gene regulatory proteins control cell multiplication and instruct cells to adopt distinct cell fates. Dysregulation of these master switches may affect differentiation, which can lead to “confused” cells as the potential progenitors of leukemia. We explore molecular mechanism that govern these processes in homeostasis and during disease.
Currently, we concentrate on the functions of “pioneering transcription factors” CCAAT Enhancer Binding Proteins alpha and beta (C/EBPα,β) in development and disease. Both C/EBPs command and control molecular programs of proliferation and differentiation into innate immune cells, including various types of myeloid cells. Previously, we have shown that dysregulation of the WNT-signaling pathway and interference with the transcription factor IRF8 are major deregulated pathways in chronic myelogenous leukemia (CML).
We apply targeted mouse genetics in combination with experimental hematopoiesis, genetic engineering of hematopoietic cells, human iPSC hematopoietic differentiation, proteomics and biochemistry. Together with the associated research group of Dr. Alexander Mildner we perform ATACseq, ChIPseq and single cell analysis to disclose fundamental epigenetic mechanisms in hematopoiesis, homeostasis, autoimmune diseases, and in leukemogenesis.
Research
Hematopoietic stem cells in the bone marrow sustain their own maintenance and give rise to “transit amplifying”, lineage committed cells that terminally differentiate into distinct, mostly short-lived cell types. Hematopoietic transcription factors (= gene regulatory proteins) concertedly control cell proliferation, lineage commitment, and cell differentiation. Mutations in these key transcription factors dysregulate hematopoiesis and cause diseases, such as leukemia and immune defects. Experimental hematology, leukemia research, in combination with stem cell research strives to untangle the underlying “grammar” of transcription factor networks.
We disclose molecular and functional interactions between regulatory proteins and determine how they orchestrate the complex process of proper gene expression and thereby reveal novel therapeutic targets. The lab currently focuses on functions of transcription factors of the CCAAT Enhancer Binding Protein family (C/EBPs). We use state-of-the-art molecular genetics, targeted mouse genetics, genomic approaches and proteomics to unravel how C/EBPs connect to innate immunity and onco-developmental transformation.