Loss of control in heart muscle cells
A team of experts with scientists from the German Center for Cardiovascular Research (DZHK) has now demonstrated for the first time that many shortened and less healthy titin proteins are present in the heart muscle cells of patients with a certain mutation in the titin gene. This weakens the heart muscle and can lead to dilated cardiomyopathy. In this case, the ventricles and atria are abnormally dilated and the heart can no longer pump properly.
Until now, it was not clear whether mutations in the titin gene lead to a shorter form of the protein or whether this shortened protein is degraded.
“The causes of dilated cardiomyopathy are manifold, but clearly the most common is a mutation that shortens the titin gene,” says project leader Professor Wolfgang Linke, director of the Institute for Physiology II at the University of Münster (WWU) and leader of a working group at the Heart Center in Göttingen. Like most genes, the titin gene has two copies (alleles) in the human body. In patients with dilated cardiomyopathy (DCM) carrying the mutated titin, only one of the two titin genes is shortened, the other still has its full length and usually is healthy. “Until now, it was not clear whether mutations in the titin gene lead to a shorter form of the protein or whether this shortened protein is degraded,” says Professor Norbert Hübner, head of the Lab “Genetics and Genomics of Cardiovascular Diseases” at the Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC), who was also involved in the study.
Theoretically, it would be possible that the healthy gene could produce enough titin protein to properly supply the heart muscle cells. Using more than 100 tissue samples from human hearts, the research team found that although the healthy allele produced more normal protein than usual in DCM patients with the titin mutation, there was still significantly less normal titin protein present than in healthy hearts or those of DCM patients without the titin mutation. Less titin means less contractile units (sarcomeres) in the heart muscle cells and thus, less contractile force development. As a result, the heart becomes a weaker pump.
Quality control of the cells fails
The scientists also demonstrated for the first time that the cardiac muscle cells of DCM patients contain shortened titin proteins. Until now, these truncated proteins had not been found. “We show that the shortened titin proteins are of no use to the heart muscle cells, because they are not incorporated into the sarcomeres, so the cardiac muscle cells contain less contractile units,” Linke says. Instead, the shortened proteins collect in small particles within the cells. Similar to neurodegenerative diseases like Alzheimer’s, these clumped proteins could be ‘toxic’, according to Linke.
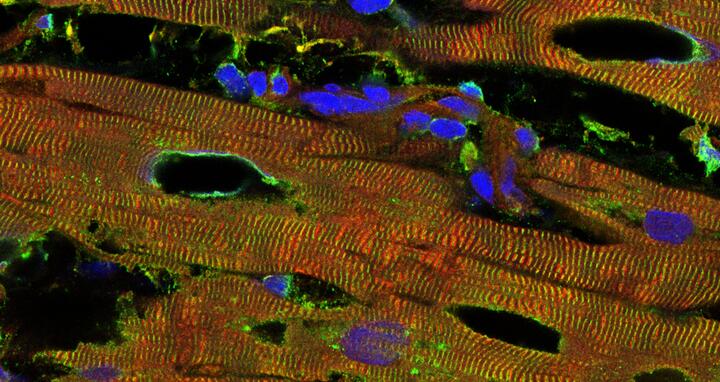
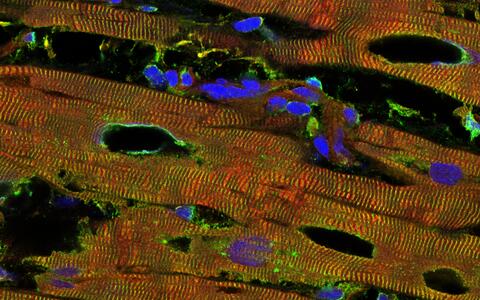
The beginning destruction of the heart tissue in an individual who has a pathologically dilated heart muscle (dilated cardiomyopathy) can be seen in black. Titin is colored red, another marker of the contractile units (alpha-actinin) is colored green, and the cell nuclei are colored blue.
The cardiac muscle cells from DCM patients with a shortened titin gene also have a problem with protein quality control systems, as the researchers recognized. Normally, mutated proteins are broken down as quickly as possible in the cells. However, the shortened titin proteins are very stable and are not recognized by the control systems of the cells.
Gene scissors can repair mutation
“Our study is groundbreaking work for the field,” Linke adds. He and his colleagues at the University Medical Center Göttingen and the Max Delbrück Center for Molecular Medicine in Berlin were also able to suggest how DCM patients with a shortened titin gene could be successfully treated or even cured. “Using heart muscle cell cultures from stem cells, we show that the CRISPR / Cas9 gene scissors can repair the mutation. To be helpful for these patients, the gene scissors would have to be applied on the exact spot of the mutated proteins, i.e. exactly on the heart muscle cell. This is not yet possible in this form — but if it becomes possible, we could cure the patients,” the project manager explains.
The study was carried out in close cooperation between the Institute of Physiology II, the Cardiology department at Münster University Hospital, the Bad Oeynhausen Heart and Diabetes Center, and the University Medical Center in Göttingen, where Linke is a visiting professor. Additional collaboration partners are the Max Delbrück Center for Molecular Medicine in Berlin and the Cardiology department at the Technical University of Munich. The work was supported by the German Research Foundation (Collaborative Research Center 1002), two funding lines from the Medical Faculty of Münster (IZKF and MedK) and the German Center for Cardiovascular Research.
Text: DZHK, Universität Münster
Further information
- Press release: “Unknown mini-proteins in the heart”
- German Center for Cardiovascular Research
Literature
Andrey Fomin et al. (2021): „Truncated titin proteins and titin haploinsufficiency are targets for functional recovery in human cardiomyopathy due to TTN mutations“. Science Translational Medicine, DOI: 10.1126/scitranslmed.abd3079
Picture to download
The beginning destruction of the heart tissue in an individual who has a pathologically dilated heart muscle (dilated cardiomyopathy) can be seen in black. Titin is colored red, another marker of the contractile units (alpha-actinin) is colored green, and the cell nuclei are colored blue. Photo: Linke Lab, University of Münster