Unknown mini-proteins in the heart
The human heart holds many secrets. And not just in a figurative, emotional sense; also from a rational, scientific point of view, we know surprisingly little about the function of the muscular organ that supplies every cell of our bodies with oxygen – and why it sometimes doesn’t do what it’s supposed to.
A study published in the scientific journal Cell has now shed a little more light on this most vital organ. An international team of 56 researchers led by the MDC examined the proteins produced by the ribosomes – our bodies’ cellular protein factories – in the heart cells of both healthy people and those suffering from heart disease. The results were surprising, and included the discovery of a large number of mini-proteins that were previously entirely unknown.
The work involved scientists from Berlin, including several groups from the MDC and Charité, as well as researchers from Bad Oeynhausen, Göttingen, Hamburg, Münster, Australia, the United Kingdom, Japan, the Netherlands, Singapore, and the United States.
DNA contains far more blueprints than previously thought
These proteins seem to be evolutionarily quite young. We could not find them in mouse hearts, for example.
The DNA stored in the nucleus of every cell contains a blueprint for all proteins produced in the body. The production of protein is a two-step process: transcription and translation. In the first step, copies of DNA fragments are produced in the form of messenger RNAs (mRNAs), which then leave the cell nucleus. In the second step, ribosomes use individual amino acids swimming around in the cell to create the corresponding proteins. While there has been quite a lot of scientific research into transcription, comparatively little is known about the translation process.
“With the help of a relatively new technique known as ribosome profiling, or Ribo-Seq, we have now been able to determine for the first time not only in isolated cells, but also in intact human heart tissue, which mRNA sites the ribosomes migrate to,” explains Dr. Sebastiaan van Heesch, a member of Professor Norbert Hübner’s Genetics and Genomics of Cardiovascular Diseases group at the MDC and lead author of the study. “Using special algorithms, we were then able to calculate which proteins are produced in the heart during translation.”
Using this technique, the researchers discovered a whole series of tiny, previously unknown proteins. Another surprising discovery made by van Heesch and the team was that many of the microproteins were encoded by RNAs that were not believed to have encoding properties – i.e., not thought to contain instructions for building proteins.
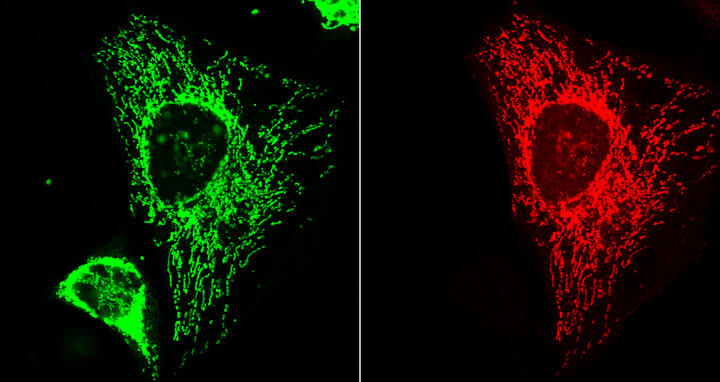
A whole series of tiny, previously unknown proteins are produced in the heart. A large portion of these microproteins migrate to the mitochondria, the cell’s energy powerhouses, after their production. The image provides proof that one of the new microproteins (red) reached the mitochondria (green). The yellow area on the third image shows that the mitochondrial signal overlaps with that of the microprotein inside the cells and that the microprotein is thus located in the mitochondria. The cell’s nucleus is blue.
Most mini-proteins are used for energy production
Using special microscopic techniques, the scientists were then able to observe that, once produced, more than half of these microproteins migrate to the mitochondria – the energy powerhouses of our cells. “This means that they are obviously used in the heart’s energy production processes,” says Norbert Hübner. “Since many heart diseases are caused by a faulty energy metabolism, we were particularly interested in this result.”
In order to detect possible differences between the translatome (totality of proteins formed) of diseased and healthy hearts, the scientists examined tissue samples from 65 patients with dilated cardiomyopathy (DCM) – a condition in which the heart muscle becomes enlarged. The samples were taken from the patients by biopsy during scheduled heart operations. The tissue of 15 healthy hearts was used for comparison.
DCM, which requires many patients to undergo a heart transplant at some point in their lives, is caused by a mutation in the titin gene – the largest and most important protein of the human heart. “As a result of this genetic mutation, a stop signal is generated in the mRNA that tells the ribosomes to finish their work before the titin has been completed,” explains van Heesch. However, not all people who carry this mutation in their DNA will actually develop DCM.
New approaches to heart disease on the horizon
Van Heesch and his colleagues are now investigating the reasons behind their discoveries. “We have observed that ribosomes can sometimes simply ignore this stop signal and continue undeterred with titin production,” says the researcher. The goal now, he explains, is to find out the circumstances under which this occurs. Van Heesch explains that it may be due to the position of the genetic mutation on the mRNA, but that it could also be the result of factors that, once identified, may be treatable.
Together with his colleagues, van Heesch also hopes to more closely investigate the role of the newly discovered microproteins. “These proteins seem to be evolutionarily quite young,” he says. “We could not find them in mouse hearts, for example.” The substances thus offer further evidence, he claims, of just how special the human heart is. Furthermore, the scientist hopes that he will one day be able to use these proteins either for the diagnosis of heart disease or as a target structure for future therapies that will be more effective than ever before in treating a disruption in the heart’s energy metabolism. bro
Further information
Original publication
Sebastiaan van Heesch et al. (2019): “The Translational Landscape of the Human Heart.” Cell, DOI:10.1016/j.cell.2019.05.010.
- The Max Delbrück Center for Molecular Medicine (MDC)
-
The Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC) is one of the world’s leading biomedical research institutions. Max Delbrück, a Berlin native, was a Nobel laureate and one of the founders of molecular biology. At the MDC’s locations in Berlin-Buch and Mitte, researchers from some 60 countries analyze the human system – investigating the biological foundations of life from its most elementary building blocks to systems-wide mechanisms. By understanding what regulates or disrupts the dynamic equilibrium in a cell, an organ, or the entire body, we can prevent diseases, diagnose them earlier, and stop their progression with tailored therapies. Patients should benefit as soon as possible from basic research discoveries. The MDC therefore supports spin-off creation and participates in collaborative networks. It works in close partnership with Charité – Universitätsmedizin Berlin in the jointly run Experimental and Clinical Research Center (ECRC), the Berlin Institute of Health (BIH) at Charité, and the German Center for Cardiovascular Research (DZHK). Founded in 1992, the MDC today employs 1,600 people and is funded 90 percent by the German federal government and 10 percent by the State of Berlin.