The metamorphosis of a chloride ion channel
Ion channels have vital functions: They ensure that ions such as chloride, potassium or sodium can flow in and out of our cells. Thereby they regulate the electrolyte content of cells and their environment and can generate electrical signals. Ion channels are also very important for the function of intracellular organelles. ASOR is a pH sensitive chloride channel that is present both in outer and inner membranes of cells.
Until now, little has been known about the Acid-Sensitive Outwardly Rectifying Anion Channel – ASOR for short. In 2019, the team of Professor Thomas Jentsch from the Leibniz Research Institute for Molecular Pharmacology (FMP) and Max Delbrück Center for Molecular Medicine (MDC) in Berlin identified the gene for the acid-sensitive ion channel, in parallel with another group in the USA. Long before, it was known that this channel only opens when the extracellular environment becomes very acidic. This is unusual, because such a low pH normally only occurs when cells die – for example, in case of stroke or heart attack.
To date, it is unclear why virtually all human and animal cells possess this ion channel. Recent data suggest that ASOR plays an important role in intracellular vesicles, whose pH is acidic enough to activate ASOR. However, the mechanism of this activation, and the structure of the chloride-conducting pore were unknown. Knowledge of these properties is, however, a prerequisite for designing pharmaceuticals that may affect ASOR. In close collaboration with structural biologists in New York, the Berlin researchers have now gained important new insights: For the first time, the structure of the open channel was shown and a novel activation mechanism was identified.
Structure of the open channel described
Using cryo-electron microscopy, the collaborating team led by Professor Steve Long at the Memorial Sloan Kettering Cancer Institute in New York, obtained high-resolution structures of the channel at different pH values. Models for the molecular mechanisms underlying ASOR’s function were derived from a comparison of these structures. They were tested by the Berlin team using replacements of single amino acids and subsequent electrophysiological analysis.
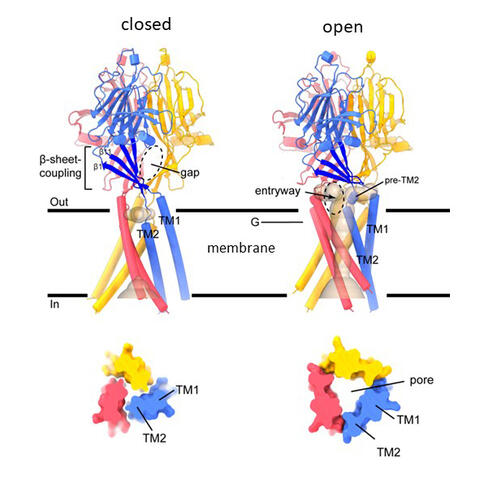
“What we found is quite unusual for an ion channel,” says postdoctoral researcher Maya Polovitskaya, one of the study’s first authors. “When pH changes the channel’s extracellular domain contract, which thereby pulls on membrane-spanning helical segments of the channel protein. Unlike other channels, where opening of the pore involves only relatively small changes in the position of a single or a few amino acids, we see a dramatic change in the membrane-spanning segment of ASOR. We call this process transmembrane metamorphosis. It results in the formation of a pore through which chloride flows. This remodeling is strikingly different from the opening mechanism of other known channels.”
How the metamorphosis of the channel is triggered
Structure of the ASOR channel in the closed and open acid-activated configuration. ASOR consists of three identical TMEM206 proteins. Each protein spans the membrane twice. Upon opening, the protein undergoes significant restructuring. Thus, a central pore (shown in light brown, top right) is formed through which the chloride ion can pass. A cross-section at level G — inside the membrane — is shown below. Figure: Excerpts from Figure 2
By comparing the channel structures at neutral and acidic pH, the researchers also tracked down the underlying activation mechanism. For each subunit of the channel, which is composed of three identical proteins, there are three pairs of negatively charged amino acids in the extracellular domain. They normally repel each other electrically. At acidic pH, i.e., a high concentration of H+ (protons), these positively charged particles intercalate between the negative side chains of the amino acid pairs and ‘stick’ them together. This results in the contraction of the extracellular part described above and the formation of the membrane pore. This mechanism explains the strong pH dependence of ASOR.
The sodium channel ASIC, known for decades, is also opened by acidic pH. However, the mechanism of pH sensitivity and the iris-like opening of ASIC’s pore are fundamentally different from ASOR. “In our work, we have discovered new mechanisms and laid a foundation for the development of ASOR-influencing compounds,” says Polovitskaya.
The question remains: what is ASOR actually good for? Some time ago, a group from the USA showed that the ion channel plays a fatal role in stroke. Knock-out mice in which ASOR was switched off survived the stroke with less permanent damage than their counterparts with a functioning channel.
Cell death may not be the only purpose of ASOR
“ASOR definitely plays a role in acid-induced cell death, but a role in intracellular processes, for example in the transport of vesicles, is now coming to the fore,” says Thomas Jentsch. He has already discovered a number of ion channels, described their biological functions and showed that some of them are mutated in human disease. The researcher is confident that he will also be able to solve the mystery of ASOR. “We already have evidence that inhibiting the channel could mitigate brain cell death in stroke. But being basic researchers, we naturally also want to understand the biological function in healthy organisms,” Jentsch says. “Much of our effort is directed towards solving this exciting question.”
Text: FMP
Further information
Literature
Chongyuan Wang, Maya M. Polovitskaya et al. (2022): „Gating choreography and mechanism of the human proton-activated chloride channel ASOR“. In: Science Advances, DOI: 10.1126/sciadv.abm3942