The mechanisms behind swallowing
Swallowing disorders can have many causes, and they occur more frequently in older people. But neurological diseases such as multiple sclerosis and Parkinson’s disease, and certain medications, can also prevent food moving normally from mouth to stomach. Possible consequences include malnutrition, weight loss, and dehydration.
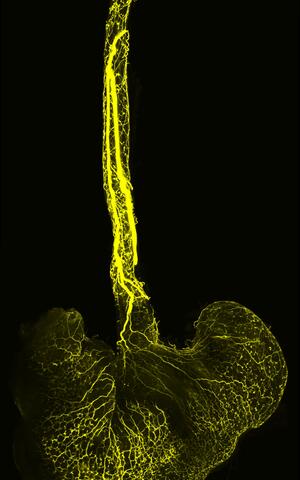
Image of the mouse’s esophagus and stomach: The team labeled the axonal projections of the sensory neurons originating in a ganglion of the vagus nerve (vagal ganglion) with a fluorescent dye. A mesoSPIM microscope (light-sheet microscopy) then made the axons visible.
Now a team led by Professor Carmen Birchmeier, who runs the Developmental Biology/Signal Transduction Lab at the Max Delbrück Center in Berlin, has investigated the process of swallowing in more detail. Writing in the journal “Neuron”, the researchers describe how sensory cells of the vagus nerve react to mechanical stimuli in the esophagus and trigger involuntary muscle movements, a process known as esophageal peristalsis. The vagus nerve, one of the 12 cranial nerves, provides information about the status of inner organs to the brain. The team’s study results could eventually lead to better treatments for swallowing disorders.
Swallowing on camera
“Modern methods of single-cell sequencing made our work possible,” explains Birchmeier. “Using the sequencing data, we constructed genetic models that allowed us to study the functions of the sensory neurons in the vagal ganglia in more detail.” Ganglia are a group or “node” of neuronal bodies in the peripheral nervous system.
The scientists began by staining the neurons to see which organs they innervate. Then they determined whether and how they responded to mechanical stimuli in the esophagus. Lastly, they deactivated the cells to analyze how this affected swallowing. Dr. Teresa Lever of the University of Missouri School of Medicine in Columbia, USA, developed a method that allowed the researchers to use video fluoroscopy to observe the swallowing in freely behaving, non-anesthetized mice in real time.
More than just a tube
“When mice lost the neurons that provide information about mechanical stimuli in the esophagus, they lost the ability to reflexively perform the appropriate muscle movements that transport food to the stomach, and they quickly lost weight,” says lead author Dr. Elijah Lowenstein, who earned his PhD working on this study in Birchmeier’s team. He’s now a researcher at Harvard Medical School in Boston. The weight loss, says Lowenstein, shows that the neurons play a key role in bodily homeostasis.
“So, the esophagus isn’t just a tube that connects the mouth to the stomach,” he says. “It uses mechanosensory feedback to fulfill its function.” Birchmeier adds that without these cells in the vagus nerve, food gets stuck in our esophagus. In some of the mice, it actually flowed back into the throat.
A molecular atlas for all
“Our work can now help develop better treatments for swallowing disorders. One option would be to pharmacologically activate the mechanoreceptors we identified,” says Birchmeier. She also wants to use the genetic models to determine the functions of other vagal sensory neurons – such as those that control the lungs or aorta.
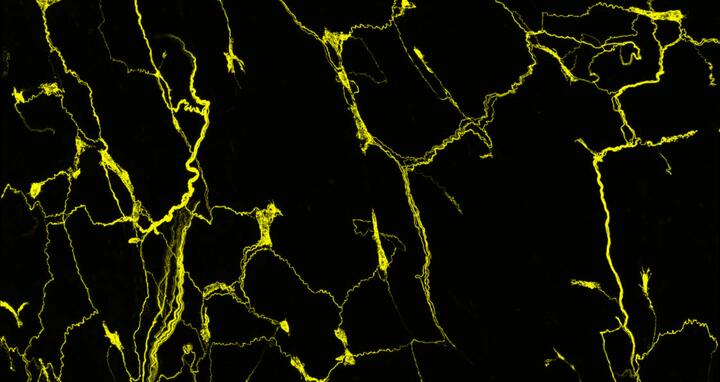
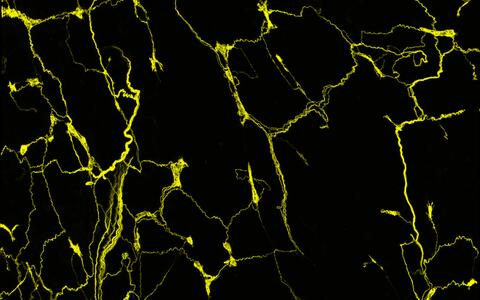
The axons of the sensory neurons form an intricate network in the mouse esophagus. The axonal projections of the sensory neurons, which originate in a ganglion of the vagus nerve (vagal ganglion), are stained with a fluorescent dye. A confocal microscope renders them visible.
“These neurons probably play a crucial but as-yet unknown role in the development of certain respiratory diseases, or cardiovascular diseases such as hypertension,” she says. Other researchers can also participate in these projects, as Birchmeier and her team have developed a molecular atlas for all vagal neurons in mice. The atlas is freely available online.
Text: Anke Brodmerkel
Further information
Literature
Elijah D. Lowenstein et al. (2023): „Prox2 and Runx3 vagal sensory neurons regulate esophageal motility“. Neuron, DOI: 10.1016/j.neuron.2023.04.025
Downloads
Image of the mouse’s esophagus and stomach: The team labeled the axonal projections of the sensory neurons originating in a ganglion of the vagus nerve (vagal ganglion) with a fluorescent dye. A mesoSPIM microscope (light-sheet microscopy) then made the axons visible. Photo: Elijah D. Lowenstein, C. Birchmeier Lab, Max Delbrück Center
The axons of the sensory neurons form an intricate network in the mouse esophagus. The axonal projections of the sensory neurons, which originate in a ganglion of the vagus nerve (vagal ganglion), are stained with a fluorescent dye. A confocal microscope renders them visible. Photo: Elijah D. Lowenstein, Birchmeier Lab, Max Delbrück Center
Contacts
Prof. Dr. Carmen Birchmeier
Head of the Lab “Developmental Biology / Signal Transduction”
Max Delbrück Center
+49 (0) 30 9406 – 2403
cbirch@mdc-berlin.de
Jana Schlütter
Editor, Communications
Max Delbrück Center
+49 (0)30 9406 – 2121
jana.schluetter@mdc-berlin.de or presse@mdc-berlin.de
- Max Delbrück Center
-
The Max Delbrück Center for Molecular Medicine in the Helmholtz Association (Max Delbrück Center) is one of the world’s leading biomedical research institutions. Max Delbrück, a Berlin native, was a Nobel laureate and one of the founders of molecular biology. At the locations in Berlin-Buch and Mitte, researchers from some 70 countries study human biology – investigating the foundations of life from its most elementary building blocks to systems-wide mechanisms. By understanding what regulates or disrupts the dynamic equilibrium of a cell, an organ, or the entire body, we can prevent diseases, diagnose them earlier, and stop their progression with tailored therapies. Patients should be able to benefit as soon as possible from basic research discoveries. This is why the Max Delbrück Center supports spin-off creation and participates in collaborative networks. It works in close partnership with Charité – Universitätsmedizin Berlin in the jointly-run Experimental and Clinical Research Center (ECRC), the Berlin Institute of Health (BIH) at Charité, and the German Center for Cardiovascular Research (DZHK). Founded in 1992, the Max Delbrück Center today employs 1,800 people and is 90 percent funded by the German federal government and 10 percent by the State of Berlin.