Zampieri Lab
Development and Function of Neural Circuits
Profile
Team
Research
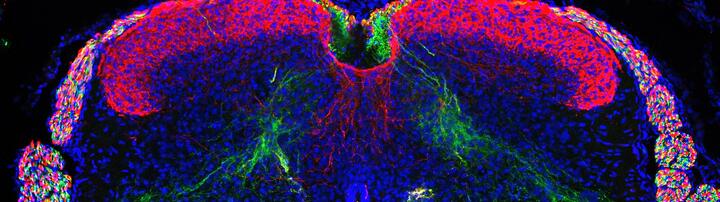
The sixth sense, proprioceptive sensory neurons and circuits controlling the sense of body position in space
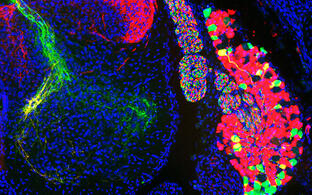
The execution of coordinated movements depends on proprioception, the sense of body position in space. The main source of proprioceptive information in vertebrates is represented by specialized mechanoreceptive organs — muscle spindles and Golgi tendon organs — found in skeletal muscles. These structures are innervated by proprioceptive sensory afferents and detect muscle stretch and tension. The central nervous system integrates this information in order to generate a three-dimensional model of the body that is used to plan, execute, and adjust movements. Despite the importance of muscle sensory feedback for motor control the organization and function of different proprioceptive circuits is still not clear. We used a transcriptomic approach to unravel the molecular foundation of mouse proprioceptor subtype identity and discovered signatures that define proprioceptors innervating different muscle groups (Dietrich et al., Nat. Commun. 2022), by leveraging this knowledge we are designing a genetic toolbox to target different components of the proprioceptive system and define their roles in the control of movement.
An intraspinal mechano and chemosensory system for motor control
A century ago, Kolmer and Agduhr first described a peculiar population of spinal sensory neurons lining the central canal and extending a ciliated protrusion into its lumen. Because of their morphology, anatomical location, and conservation in all vertebrates, they proposed that cerebrospinal fluid-contacting neurons (CSF-cN) constitute an internal sensory organ relaying chemical and mechanical information from the CSF. We analyzed CSF-cN function in mice and found specific defects in skilled locomotion suggesting the hypothesis that CSF-cN monitor spinal bending and provide an additional source of proprioceptive information that is used to precisely adjust body and limbs movements (Gerstmann et al., Curr. Biol. 2022). We seek to define the type of sensory information relayed by CSF-cN, how it is detected and integrated with other sensory feedback systems, and its specific contributions to motor control.
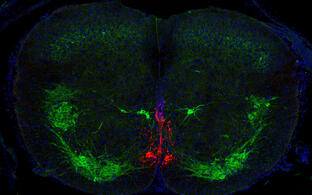
The organization and function of spinal circuits for integration of sensorimotor information
A critical aspect of motor control is the seamless coordination in the movement of different parts of the body. Limbs and trunk movements have to be regulated in a concerted fashion to allow the generation of complex motor actions. At spinal level, this important task is controlled by propriospinal neurons. These neurons resides and connect within the spinal cord, forming circuits that wire different segmental levels, thus providing channels for exchanging motor and sensory information to coordinate and adjust forelimb, hindlimb and trunk movements. While the existence and importance of propriospinal neurons has been appreciated for over one hundred years, the precise composition and roles of these circuits are still not known yet. By combining anatomical tracing techniques, transcriptome analysis and functional interrogation at a behavioral level, we seek to define molecular idenitity, connectivity and function of propriospinal neurons.
Publications
News
Lab
At the Deutsches Hygiene-Museum in Dresden to talk about proprioception to children and families
Elisa’s defense 2025
Motor Symposium FENS 2024
X‑mas bowling 2023
Georgian dinner 2023
X‑mas glühwein 2022
Bowling July 2022
Lab picture April 2021
Lab dinner July 2020
Lab picture March 2019
Lab dinner November 2018
Lab picture September 2018
Tiergarten 5X5 relay 2017
Lab dinner October 2016
Lab picture April 2016
Lab dinner November 2015