Histone H3-tail positioning and acetylation by the c‑Myb but not the v‑Myb DNA binding SANT domain
The production of blood cells, including erythrocytes, granulocytes, macrophages, and immune cells, is controlled by nuclear regulatory proteins that determine transcription of distinct genes from DNA. Several of these transcription factors, normally instrumental for balancing hematopoietic proliferation versus cell differentiation, can turn into leukemia genes by mutations.
One of these critical transcription factors essential for hematopoietic development is Myb. Myb is essential for the expansion of precursor cells of all blood cell lineages. Its name was derived from a leukemia virus with the ability of the virus to induce myeloblastosis. The myeloblastosis virus, isolated about 70 years ago, carries a recombinant version of the Myb gene that has been captured from the cellular Myb gene. The cellular Myb gene product balances proliferation, differentiation and controls lineage fate in a concerted fashion together with proteins of the C/EBP transcription factor family. In contrast, viral Myb has been turned into a leukemia gene by mutations which block myeloid cell differentiation and which induce proliferation. Hence, determining the differences between viral and cellular Myb apart will shed light on how normally cell lineage fate is accomplished and how it is disrupted in leukemia.
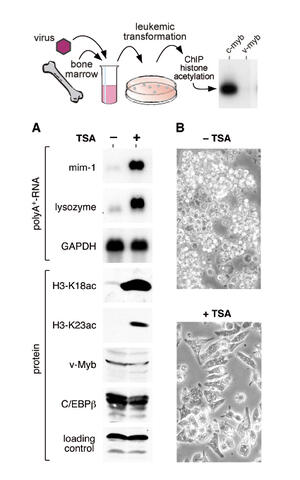
The cellular Myb protein has three consecutive N‑terminal SANT-type repeat domains (R1, R2, R3), two of which (R2, R3) form the DNA binding domain. Three amino acid substitutions in R2 of the viral Myb protein alter the way Myb regulates genes and determine the leukemogenicity of the retrovirally transduced v‑Myb oncogene. The molecular mechanism of how these mutations unleash the leukemogenic potential of Myb, has remained unknown for a long time, however. Now, Xianming Mo and collaborators (laboratory of Prof. Achim Leutz) were able to demonstrate that the c‑Myb DNA binding domain interacts with the N‑terminal histone tails of H3 and H3.3. C‑Myb binding facilitates histone tail acetylation, mandatory during activation of major differentiation genes in conjunction with CCAAT enhancer binding proteins (C/EBP) (Genes and Development doi: 10.1101/gad.355405). The leukemogenic mutations in v‑Myb R2 eliminate the interaction with H3 and prevent acetylation of H3 tails thereby abolishing activation of differentiation genes. It was demonstrated that pharmacologic enhancement of H3 acetylation restored activation of differentiation genes and induced cell differentiation in primary v‑myb transformed myeloblasts. These data establish a link between a novel, epigenetic chromatin function of c‑Myb with lineage specific expression of differentiation genes and relates the loss of the epigenetic function with leukemic conversion.
Contact:
Pamela Cohen
p.cohen@mdc-berlin.de
+49 30 9406 2121