Working the springs and pistons of the heart
A new study by Michael Gotthardt and colleagues at the Max-Delbrück Center and Washington State University shows that titin is not just a passive spring; it also receives signals that govern the contraction of muscles and are crucial to the health and proper function of the heart. The findings, published in the current issue of Circulation, help explain why people who inherit a defective form of titin develop cardiac disease or muscular dystrophy.
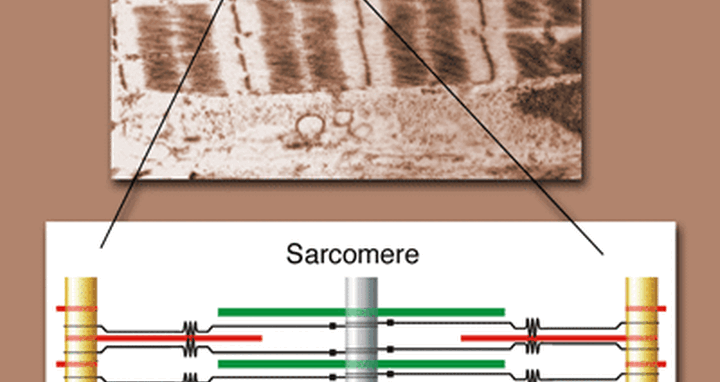
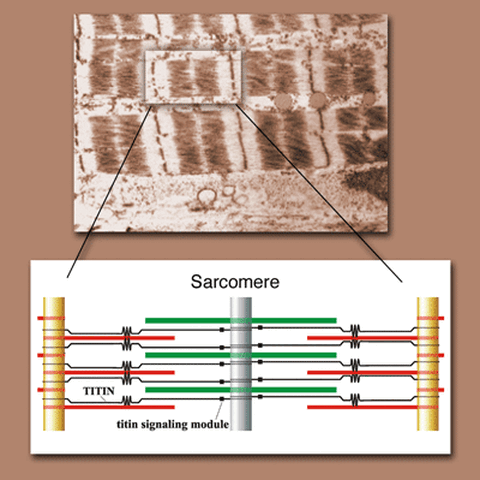
Above: A microscope image of sarcomeres. One sarcomere is highlighted. Below: A graphic representation of one sarcomere, showing the position of titin (black) and its signaling module (black square). The piston-like movement of the sarcomere occurs when thin protein filaments (red) slide in and out along thick filaments (green). _Note: Sarcomere graphic copyright 2007 by Michael Gotthardt ._
The muscle that makes up the heart is composed of long rows of piston-like units called sarcomeres. Titin is anchored to the ends of each sarcomere and connects all the pieces, giving the structure its elasticity. Yet the protein clearly does more – mutant forms of titin have been found in patients suffering from cardiac and muscle disease. Additionally, a region within titin enables it to process signals from other proteins. One hypothesis suggested that this module might act as a sensor for the amount of stress placed on the sarcomere, but its exact function was unknown. Gotthardt and his colleagues wondered what would happen if signals passed through titin were blocked.
To find out, they developed a strain of mouse in which titin’s signaling module could be removed. One way to achieve this would be to develop a “knockout” animal without the module at all, but such animals die early in embryonic development. Investigating the effect of titin signaling in the adult heart required a more sophisticated technique called a “conditional knockout,” in which a protein’s function can be shut down at any time.
Switching off titin signaling in adult mice led to severe problems. The piston-like structure of sarcomeres began to dissolve, and heart muscle no longer contracted well. Gotthardt and his colleagues traced the effect to a dramatic drop in cells’ ability to move calcium, which is crucial in coordinating muscle activity. The animals’ hearts also became enlarged due to an accumulation of fibers. Eventually the damage proved fatal.
Titin has been known to help muscle tissue relax; this is the first time the molecule has been connected to muscular contraction and cells’ handling of calcium. It shows that titin plays a much more complex role in controlling the behavior of sarcomeres and the heart than previously thought. This helps account for cardiac disease and muscular dystrophy that arise because of mutations in titin, and it gives medical researchers a new place to start as they look for treatments.
Russ Hodge
Highlight Reference:
Circulation, Jan. 29, 2007 (online) Cardiac Hypertrophy and Reduced Contractility in Hearts Deficient in the Titin Kinase Region Jun Peng, Katy Raddatz, Jeffrey D. Molkentin, Yiming Wu, Siegfried Labeit, Henk Granzier and Michael Gotthardt Department of Veterinary and Comparative Anatomy, Pharmacology, and Physiology, Washington State University, Pullman (J.P, Y.W., H.G., M.G.); Neuromuscular and Cardiovascular Cell Biology, Max-Delbrück Center for Molecular Medicine, Berlin, Germany (K.R., M.G.); Division of Molecular Cardiovascular Biology, Department of Pediatrics, Children’s Hospital Medical Center, Cincinnati, Ohio (J.M.); and Department of Anesthesiology, Mannheim University, Mannheim, Germany (S.L.).
An excellent review on titin from authors Henk Granzier and Siegfried Labeit:
The Wikipedia entry on titin:
An article about Henk Granzier’s receipt of the Helmholtz Humboldt Research Award:
full version of the article at Circulation