Anchors aweigh on an autoimmune disease
The watchmen of the immune system – white blood cells, or leukocytes – squeeze through the lining of blood vessels to seek out invaders in the body’s tissues. When they find one, leukocytes are activated in a burst of substances that destroy invading particles or cells. In systemic vasculitis, that activation happens too early, injuring vessels and eventually destroying the organs that they supply with blood. One form of vasculitis is an auto-immune disease that occurs when the body builds antibodies (called ANCA) against proteins which appear on the leukocytes themselves. Moving from patients to the lab, the team of Ralph Kettritz and Friedrich C. Luft (at the Charité/HELIOS university clinic and the MDC) have discovered that two proteins work together to trigger inflammations in this disease. Their study appears in the on-line edition of the journal Blood.
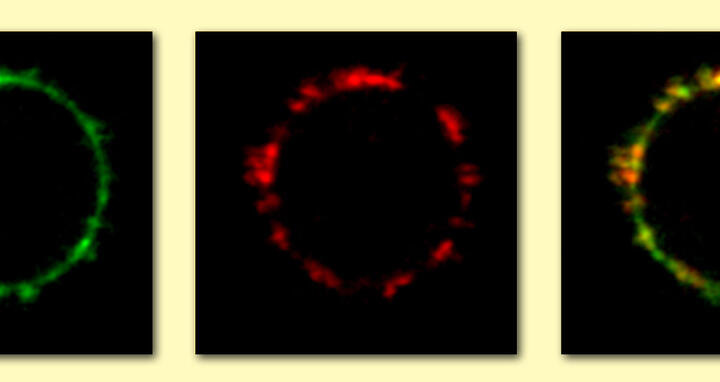
Sibylle von Vietinghoff and other members of the team knew that ANCA-vasculitis patients develop antibodies against a protein called PR3, which is produced only in leukocytes. The body uses antibodies to mark objects that are likely to be foreign and dangerous, calling them to the attention of the immune system. Sometimes – as in the case of PR3 – antibodies target one of the body’s own proteins and the immune system begins to attack things it shouldn’t. Most PR3 is kept out of reach, glued to internal compartments of leukocytes, but occasionally it appears on the outer surface of the cells. In ANCA-vasculitis, much more of it moves from the interior to the surface. If the body builds antibodies against it, the result is a dangerous chain of inflammation that can become deadly.
Current therapies against the disease shut down the body’s ability to manufacture a wide range of antibodies, including those for PR3, but this aggressive tactic weakens the body’s defenses against other diseases. Vietinghoff and her colleagues thought that something might be learned from the unusual surface migration of PR3. They compared blood cells from healthy people to those of patients with ANCA-vasculitis. As expected, the patients had more PR3 on the surface of leukocytes. Then a chance discovery with a patient suffering from another disease prompted the scientists to take a look at another protein called NB1. Both molecules were behaving the same way; in ANCA-vasculitis, NB1 was also moving in higher amounts to the membrane, where it formed the same pattern as PR3.
This suggested that the proteins might need to come in contact with each other to cause problems, but was that really the case? Interfering with NB1 might give some answers. The NB1 protein is anchored in the cell membrane by a particular kind of lipid – one of the small fat molecules that are the main components of membranes. Kettritz’s laboratory used an enzyme to cut the lipid, casting off the anchor and releasing NB1. Without this protein, PR3 disappeared as well.
The researchers knew of another blood disease caused by defective lipid anchors, which suggested a second test. The cells of those patients, they reasoned, would lose their NB1, just as if the anchor had been cut. The cells from one such patient had no traces of either NB1 or PR3. A third experiment provided another confirmation of the link between the two molecules: when the researchers added NB1 to leukocytes that didn’t produce any, suddenly both NB1 and PR3 appeared on the surfaces of the cells.
ANCA-vasculitis is a rare disease, but a very serious one, and finding a connection between PR3 and NB1 and its lipid anchor gives scientists new starting points to look for treatments. Autoimmune diseases take advantage of vital bodily systems that cannot simply be shut down by drugs. Therapies will likely have to be based on the fundamental causes within cells. Finding them and translating such basic discoveries into practice in the clinic will require teams like those of Kettritz and Luft, equally at home in both worlds.
Russ Hodge
Highlight Reference:
NB1 mediates surface expression of the ANCA antigen proteinase 3 on human neutrophils Sibylle von Vietinghoff, Gisela Tünnemann, Claudia Eulemnberg, Maren Wellner, M. Cristina Cardoso, Friederich C. Luft, and Ralph Kettritz
A review article about luekocytes by author Friederich Luft:
The Wikipedia article on vasculitis:
Link to the article in the journal Blood: