The inner workings of a protein shredder
Sommer and his colleagues have been focusing on the way cells check about a third of their proteins – those that are to be secreted from the cell or embedded in its membranes. As such molecules are synthesized, they are threaded into a labyrinthine structure called the endoplasmic reticulum (ER) and are checked by quality control mechanisms. Researchers have identified a number of components of this system, including proteins called E3 enzymes. But much remains to be learned about how these molecules assemble into machines that identify defective proteins and trigger their destruction.
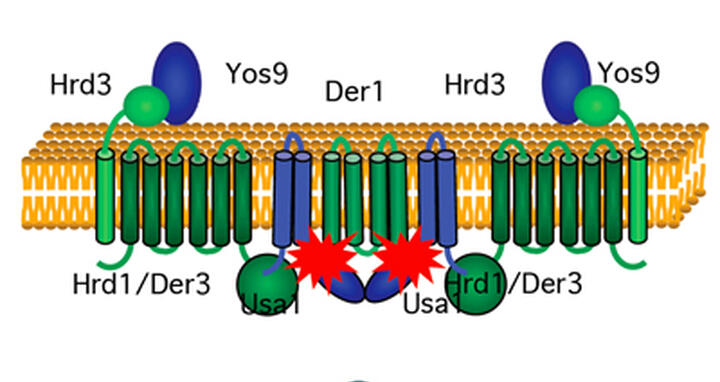
Two copies of Usa1 bind to each other to draw in modules containing Hrd1, Hrd3, and Yos9 proteins, making a scaffold for the binding of Der1. The resulting machine plays a central role in the cell’s detection of misfolded proteins.
One of the main questions concerns the flexibility of the machines: they are built from a limited number of molecules, but can still discriminate between many types of healthy and defective proteins. Part of the answer lies with “adaptor” molecules, which work a bit like the adaptor plugs needed to cope with foreign electrical outlets. On one side they plug into the machine, and the other latches onto particular types of misfolded molecules.
Experiments by PhD student Sabine Horn and other members of Sommer’s lab, teaming up with members of the Heinemann lab, have now revealed crucial aspects of the assembly and functions of one of the machines. The researchers began by investigating an E3 enzyme called Hrd1, a central component of a protein complex which sits in the membrane of the ER and scans molecules as they arrive. A number of adaptor molecules dock onto the machine to scrutinize folding. These include the proteins Usa1 and Der1, whose functions have been unclear.
“What’s interesting is that the machine is put together differently to handle various partners,” Sommer says. “Imagine that your car had to rebuild itself to drive on different kinds of surfaces. And that it could do this purely mechanically, without the aid of some sort of central computer. The machinery that detects misfolding self-assembles in different ways to handle different targets, and we’re trying to understand how it does so.”
Horn and her colleagues deleted single machine components from yeast cells – which bear strong similarities to human cells – to study the functions of the individual parts. Removing Der1 caused one group of misfolded proteins to be processed more slowly than usual, but another group was still destroyed.
Previous work hinted that Usa1 is an adaptor that helps plug Der1 into the protein complex containing Hrd1. The current study proves that this is the case, but also reveals that Usa1 has additional functions. It helps the complex recognize targets that don’t involve Der1. More importantly, it helps assemble the machine. When Usa1 binds, it draws multiple copies of Hrd1 together and likely establishes links to other components . This is necessary for the recognition and destruction of targets.
“The results strongly imply that Usa1 acts as a ‘scaffold’ for the construction of the machine,” Sommer says. “It helps keep things organized and plugs in individual components that are needed to deal with different proteins. Without this essential step, a lot of misfolded molecules slip past, and some of them go on to cause disease. This reveals a critical part of the complex that may also be a weak point as it carries out its quality control functions.”
- Russ Hodge
Highlight Reference:
Horn SC, Hanna J, Hirsch C, Volkwein C, Schütz A, Heinemann U, Sommer T, Jarosch E. Usa1 functions as a scaffold of the HRD-ubiquitin ligase. Mol Cell. 2009 Dec 11;36(5):782 – 93.
Link to the full article
Wikipedia article on the E3 ubiquitin ligase