How a mutation harms the heart
When the cardiac muscle is abnormally enlarged, the heart can no longer pump properly. It beats irregularly, causing difficulty breathing, edema, and exhaustion. The heart gradually becomes so weak that the person dies. In some families this condition, known as dilative cardiomyopathy (DCM), occurs in several members, and the gene for the protein lamin A/C is often altered in patients. But what does a protein that forms the cell’s nuclear envelope have to do with the heart’s left ventricle? A research team led by Joseph Wu and Jaecheol Lee from Stanford University answered this question in a recent paper published in the journal Nature. Dr. Sebastian Diecke, who heads the Pluripotent Stem Cell Platform at the Max Delbrück Center for Molecular Medicine (MDC) and the Berlin Institute of Health (BIH), was one of the paper’s three lead authors. He was until 2014 a postdoctoral researcher in Professor Wu’s lab, and afterwards saw the study through to publication.
Most other studies only address certain aspects of a disease. Here we were able to show how the mutation triggers the disease – using stem cells and patient tissue. In the end, we even identified a target through which certain drugs can alleviate arrhythmias.
“Most other studies only address certain aspects of a disease. Here we were able to show how the mutation triggers the disease – using stem cells and patient tissue. In the end, we even identified a target through which certain drugs can alleviate arrhythmias,” says Diecke. But it was a long road to get to this point.
Patient-specific stem cells
Seven members of an affected family – five of them sick and two healthy – provided the team with skin and blood samples. The researchers first reprogrammed these cells into induced pluripotent stem cells (iPS cells) and then differentiated them into heart muscle cells (cardiomyocytes). The cells of the five patients with an altered gene for lamin A/C also beat irregularly in the petri dish. Electrophysiological tests showed that the calcium homeostasis of these cells had become out of sync.
The researchers used genome editing to check whether the gene for lamin A/C was actually responsible for the arrhythmias. The inserted a normal variant of the gene in the DNA of the diseased cells and a variant with patient-specific mutations in the DNA of healthy cells. The effect was clear, but these experiments did not clarify the underlying mechanism.
A deformed nuclear envelope is discovered
In the next stage of the study, the team therefore took a closer look at the nuclear envelope of the diseased cardiomyocytes. They found that the cells were not forming enough lamin A/C and the nuclei no longer appeared round, but had significant indentations. This deformation could influence how tightly the two-meter-long DNA strand is packed into the cell nucleus and which DNA sections are read. The scientists therefore compared the gene expression in the healthy and diseased cells and found almost 240 genes that were more strongly activated in the diseased heart muscle cells.
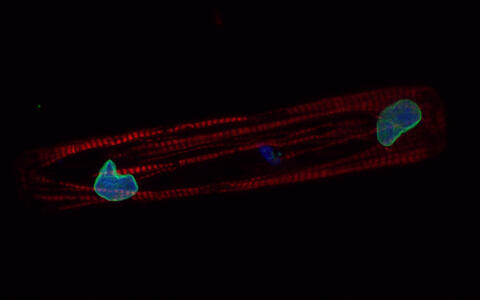
Representative confocal images of control and mutant lines. Micro-patterned iPSC-CMs were stained with specific antibodies against TNNT2 (red), and LMNB1 (green).
Many of these genes pointed to the signaling pathway for the platelet-derived growth factor (PDGF). This substance controls blood vessel formation, among other things, and is active during the differentiation of heart muscle cells, but should be largely inactive in an adult healthy heart. It is only reactivated under stress, and is associated with fibrosis. "During the study, a patient from the family we were examining had to get a pacemaker. This provided us with an additional sample of his heart tissue," reports Diecke. The same genes were expressed more often in the patient’s tissue than in the diseased cardiomyocytes cultured in the petri dish. It was also shown that PDGF controls a protein that regulates the cells’ calcium homeostasis.
A new area of application for approved cancer drugs?
The team used crenolanib and sunitinib, two cancer-fighting agents, to see if they had found the cause of the dangerous disease. These inhibitors bind to a PDGF receptor and thus block the signaling pathway. The heart muscle cells treated in this way did indeed start beating regularly again – even though the missing lamin A/C and the deformed nuclear envelope remained unchanged.
“Perhaps it will be possible to treat patients with dilated cardiomyopathy using already approved cancer drugs such as sunitinib, sorafenib, or axitinib,” says Diecke. But first the correct dosage needs to be established, as the active substances in these drugs have an undesirable side effect in cancer patients: they can damage the heart. “But if we know the relevant docking sites, we can of course also search for a safer alternative. It’s a real chance – and I'm glad I was part of this team.”
Text: Jana Schlütter
Further information
Literature
Jaecheol Lee, Vittavat Termglinchan, Sebastian Diecke et al. (2019): “Activation of PDGF pathway links LMNA mutation to dilated cardiomyopathy.” Nature, doi: 10.1038/s41586-019-1406-x.