New multinational study reveals titin gene mutations affect heart function in healthy individuals
Press Release by the National Heart Centre Singapore (NHCS)
The finding may help scientists to understand a paradox: namely that around one per cent of the world’s population carry this genetic mutation with no apparent effect. The key, the team now believes, is that the hearts of such people may be “primed to fail” if they suffer a second hit, whether genetic or environmental. This could mean that there are about 35 million people in this position globally. The research paper is published in leading medical journal, Nature Genetics today, 21 November 2016.
Titin is the largest protein in the human body that causes dilated cardiomyopathy, a condition in which the heart muscle becomes weakened, enlarged and cannot pump blood efficiently. Dilated cardiomyopathy is a type of inherited cardiac condition and affects about 1 in 250 people worldwide.
The researchers studied the effects of titin gene mutations in 2,495 patients with dilated cardiomyopathy. They also generated two rat models to understand the impact of these mutations on the molecular level and heart function. In addition, cardiac gene sequencing tests were performed in 1,409 healthy volunteers, coupled with 2D and 3D cardiac magnetic resonance imaging (MRI) that gave high resolution information on the heart size and shape of the study subjects. The data collected gave major new insights on multiple levels, allowing researchers to better understand the variants that represent the commonest genetic cause of dilated cardiomyopathy, yet are prevalent in the general population. The study was led by the National Heart Centre Singapore in collaboration with Duke-NUS Medical School, Medical Research Council Clinical Sciences Centre, Imperial College London and Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC).
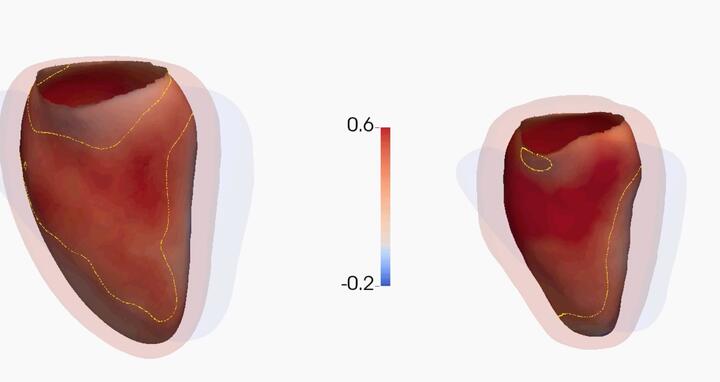
3D statistical model of the left ventricle of the heart of 1409 healthy volunteers. Left: the heart while filling with blood. Right: the heart as it contracts to pump blood around the body. In red are areas of the heart enlarged in those with titin mutations when compared to those without.
Assistant Professor Sebastian Schäfer, Senior Research Fellow at the National Heart Centre Singapore who is the first author of the paper explained: “We could directly show the impact of the mutations on the titin protein production which has an impact on the heart. Even though the heart appears healthy initially, it reacts to this genetic stress on many levels such as changes to its gene expression and energy source. The heart can compensate and its cardiac function remains fine until an additional stressor occurs. That’s when the heart fails, as it no longer has the capacity to react the same way a healthy heart does.”
Professor Stuart Cook, Tanoto Foundation Professor of Cardiovascular Medicine at the SingHealth Duke-NUS Academic Medical Centre and co-senior author, elaborated: “We now know that the heart of a healthy individual with titin gene mutation lives in a compensated state and that the main heart pumping chamber is slightly bigger. Our next step is to find out the specific genetic factors or environmental triggers, such as alcohol or viral infection that may put certain people with titin mutations at risk of heart failure.”
Dr Antonio de Marvao, Clinical Lecturer at Imperial College London and the MRC Clinical Sciences Centre, said: “Our previous work showed that mutations in the titin gene are very common in people diagnosed with heart failure. Around 1% of the general population also carry these mutations, but until now it wasn’t known if these are ‘silent’ gene changes or changes that can adversely affect the heart. Using state-of-the-art cardiac MRI, we created extremely detailed 3D “virtual hearts” from the scans of 1,409 healthy adults. We found that those with mutations have an enlarged heart, and in a pattern similar to that seen in heart failure patients. This may impact as many as 35 million people around the world. In future work we will investigate if the heart function of our volunteers is indeed impaired, by MRI scanning them as they exercise on a bike.”
Dr James Ware, Clinical Senior Lecturer in Genomic Medicine at Imperial College London and the MRC Clinical Sciences Centre, added: “For patients with dilated cardiomyopathy, this study has improved our understanding of the disease, revealed possible new targets for drugs and other new therapies, and importantly has improved our ability to diagnose the condition confidently with genetic tests. This work required a very collaborative approach, with many institutions involved in assembling genetic data from tens of thousands of individuals. The finding that titin mutations are affecting the hearts of so many otherwise apparently healthy people worldwide, and potentially increasing their risk of heart failure, poses even pressing questions, such as why some people with these mutations seem to do well in the long term, while others do not. Fortunately, we are in a strong position to tackle these questions from lots of different angles, by analysing aggregated genetic and clinical data from a network of collaborating units around the world.”
Professor Norbert Hübner, Professor of Cardiovascular and Metabolic Sciences at the MDC and co-senior author, detailed: “By using a variety of genomic approaches we showed that the RNA that is produced from the actual titin allele which carries the mutation, is degraded in the cells of the heart. This led to important insights on how these titin mutations operate.”
Currently for patients with inherited cardiac conditions, they can undergo a cardiac genetic test that will screen them of 174 genes in 17 such conditions to diagnose the exact condition and gene, to prescribe effective treatment.
The study is funded by Tanoto Foundation, National Medical Research Council Singapore, SingHealth Duke-NUS Institute of Precision Medicine, Medical Research Council Clinical Sciences Centre UK, NIHR Biomedical Research Unit in Cardiovascular Disease at Royal Brompton & Harefield NHS Foundation Trust and Imperial College London and British Heart Foundation UK, among others.
Sebastian Schafer1,2,17, Antonio de Marvao3,17, Eleonora Adami4, Lorna R Fiedler2, Benjamin Ng1, Ester Khin2, Owen J L Rackham2, Sebastiaan van Heesch4, Chee J Pua1, Miao Kui2, Roddy Walsh5, Upasana Tayal5, Sanjay K Prasad5, Timothy J W Dawes3, Nicole S J Ko2, David Sim1, Laura L H Chan1, Calvin W L Chin1,2, Francesco Mazzarotto5, Paul J Barton5, Franziska Kreuchwig4, Dominique P V de Kleijn6,7, Teresa Totman6, Carlo Biffi3, Nicole Tee1, Daniel Rueckert8, Valentin Schneider4, Allison Faber4, Vera Regitz-Zagrosek9,10, Jonathan G Seidman11, Christine E Seidman11 – 13, Wolfgang A Linke14,15, Jean-Paul Kovalik2, Declan O’Regan3, James S Ware3,5,18, Norbert Hubner4,10,16,18 & Stuart A Cook1,2,5,18 (2016): „Titin-truncating variants affect heart function in disease cohorts and the general population“ Nature Genetics.
1National Heart Centre Singapore, Singapore. 2Duke – National University of Singapore, Singapore. 3Cardiovascular and Metabolic Disorders Program, MRC Clinical Sciences Centre, Faculty of Medicine, Imperial College London, Hammersmith Hospital Campus, London, UK. 4Cardiovascular and Metabolic Sciences, Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC), Berlin, Germany. 5National Heart and Lung Institute and NIHR Royal Brompton Cardiovascular BRU, Imperial College London, London, UK. 6Department of Surgery, National University of Singapore, Singapore. 7Departments of Cardiology and Vascular Surgery, University Medical Center, Utrecht, the Netherlands. 8Department of Computing, Imperial College London, London, UK. 9Institute of Gender in Medicine, Charité Universitätsmedizin Berlin, Berlin, Germany. 10DZHK (German Centre for Cardiovascular Research), partner site Berlin, Berlin, Germany. 11Department of Genetics, Harvard Medical School, Boston, Massachusetts, USA. 12Division of Cardiovascular Medicine, Brigham and Women’s Hospital, Boston, Massachusetts, USA. 13Howard Hughes Medical Institute, Chevy Chase, Maryland, USA. 14Department of Cardiovascular Physiology, Ruhr University Bochum, Bochum, Germany. 15DZHK (German Centre for Cardiovascular Research), partner site Goettingen, Goettingen, Germany. 16Charité Universitätsmedizin, Berlin, Germany. 17These authors contributed equally to this work. 18These authors jointly supervised this work
###
About National Heart Centre Singapore
The National Heart Centre Singapore (NHCS, 新加坡国家心脏中心) is a leading specialist centre for cardiovascular diseases in Singapore and Asia. NHCS provides a one-stop comprehensive cardiovascular care in preventive, diagnostic, therapeutic and rehabilitative services for heart patients. Besides a preferred regional training and education centre for cardiac specialists and healthcare professionals, NHCS is also an active research and development centre for cardiovascular diseases, in particular its National Heart Research Institute Singapore, a joint research setup with Duke-NUS.
For more information, please visit www.nhcs.com.sg
About Duke-NUS Medical School
The Duke-NUS Medical School (Duke-NUS, 杜克-新加坡国立大学医学院) was established in 2005 as a strategic collaboration between the Duke University School of Medicine, located in North Carolina, USA and the National University of Singapore (NUS). Duke-NUS offers a graduate-entry, 4‑year M.D. (Doctor of Medicine) training programme based on the unique Duke model of education, with one year dedicated to independent study and research projects of a basic science or clinical nature. Duke-NUS also offers M.D/PhD and PhD programmes. As a player in Singapore’s biomedical community, Duke-NUS has identified five Signature Research Programmes: Cancer & Stem Cell Biology, Neuroscience and Behavioral Disorders, Emerging Infectious Diseases, Cardiovascular & Metabolic Disorders, and Health Services and Systems Research.
Duke-NUS and SingHealth have established a strategic partnership in academic medicine that will guide and promote the future of medicine, tapping on and combining the collective strengths of SingHealth’s clinical expertise and Duke-NUS’ biomedical sciences research and medical education capabilities.
For more information, please visit www.duke-nus.edu.sg
About Medical Research Council
The Medical Research Council is at the forefront of scientific discovery to improve human health. Founded in 1913 to tackle tuberculosis, the MRC now invests taxpayers’ money in some of the best medical research in the world across every area of health. Thirty-one MRC-funded researchers have won Nobel prizes in a wide range of disciplines, and MRC scientists have been behind such diverse discoveries as vitamins, the structure of DNA and the link between smoking and cancer, as well as achievements such as pioneering the use of randomised controlled trials, the invention of MRI scanning, and the development of a group of antibodies used in the making of some of the most successful drugs ever developed. Today, MRC-funded scientists tackle some of the greatest health problems facing humanity in the 21st century, from the rising tide of chronic diseases associated with ageing to the threats posed by rapidly mutating micro-organisms.
For more information, please visit www.mrc.ac.uk
About Max Delbrück Center
The Max Delbrück Center for Molecular Medicine in the Helmholtz Association was founded in January 1992 with the goal of linking basic science to clinical research. The MDC was named for Max Delbrück, a physicist, biologist, and Nobel Prize winner. Currently the institute employs more than 1600 people from nearly 60 countries; over 1300 of those are directly involved in research. The MDC’s annual budget is over 80 million Euros, along with substantial third-party funding obtained by individual scientific groups.