An ornamental fish with superpowers
Daniela Panáková exploits the zebrafish’s huge potential as an experimental animal. At the same time, it’s important to her to maintain an acceptable standard of animal welfare.
Entering the basement room, the visitor is greeted by a cosy warmth and the monotonous gurgling of water. Hundreds of small aquariums are arranged in rows and neatly stacked on top of one another. Behind blue plastic panels, fish as long as a matchstick glide back and forth. “These are our zebrafish, the starting point for all our experiments,” says Dr. Daniela Panáková, as her gaze rests on the freshwater fish. Daniela Panáková heads the Electrochemical Signaling in Development and Disease working group at the Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC). She, her research team and the animal care team spend a lot of time in this room. “During the first morning feed, the animals in all the aquariums get a visual check so that we can spot any changes in scale structure or swimming behaviour as early as possible. Both of these are signs that the fish are not doing well,” the developmental biologist explains.
The MDC researcher already knew she wanted to work with zebrafish during her doctoral degree. Asked why she decided to specialise in studying congenital heart defects and cardiovascular diseases, Panáková says with a laugh: “The heart is the key to life.” She then enthusiastically recounts the scientific tradition and explains why zebrafish are so valuable as experimental animals in cardiovascular research.
How an ornamental fish became key to science
Back in 1934, Charles Creaser described the technical requirements for using the eggs of the fish Danio rerio for experiments in developmental biology regardless of the time of year. Forty years later, biologist George Streisinger prompted a revival in the use of zebrafish in basic biomedical research. The story goes that Streisinger asked the owner of a pet shop what would be a suitable vertebrate for scientific study, to which the shopkeeper simply responded by pointing to the aquarium containing zebrafish. It was the beginning of a long-lasting relationship.
Today, more than a thousand research laboratories worldwide work with this schooling fish. But nowadays, scientists don’t need to go to a pet shop. The European Zebrafish Resource Center (EZRC) at the Karlsruhe Institute of Technology is home to a central archive including thousands of zebrafish lines and more than 80,000 frozen sperm samples. At the EZRC, the tiny striped fish – originally found in the Ganges River in Pakistan, northern India and Bangladesh – are bred, characterised and shipped to researchers.
Success isn’t just in the genes
When directly compared, humans and zebrafish share over 70% of their genome. This means that scientists can use insights gained from zebrafish to draw conclusions about the genetic causes of diseases in humans. Another useful aspect from a researcher’s point of view is the fact that the animals do not require a lot of space and are not expensive to care for. They also reproduce very quickly, the females laying up to 500 translucent eggs every week. Because the eggs develop fully outside the body, the development of the organs can be observed in a Petri dish from the very first cell division.
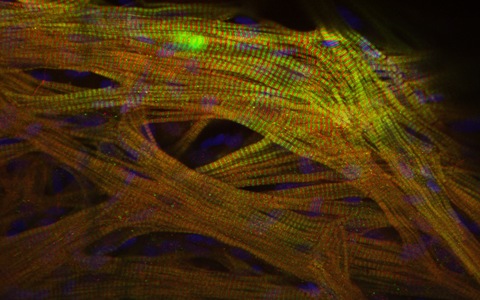
View into the heart muscle of a zebrafish
As in humans, the first functional organ in a zebrafish embryo is the heart. While the heartbeat of a human baby can be detected on an ultrasound after 29 days, the heart of a zebrafish embryo starts to contract after just 22 hours. Due to their small size, by the fifth day of development zebrafish embryos can absorb oxygen from water by means of diffusion through the body’s surface. This allows researchers to study the function of essential genes in early cardiac development. If these genes were to be switched off in an experiment with mice, for example, the animal’s life would be too severely impaired. It’s because of these traits that scientists doing basic biomedical research use zebrafish to investigate the causes of conditions such as congenital heart defects.
A heart with self-healing powers
Another reason why biomedical researchers all over the world work with zebrafish is the remarkable self-healing powers of the adult fish. Zebrafish can do something that has fascinated humanity for thousands of years and that heart attack patients can only hope for: even if up to 20% of the zebrafish heart muscle is removed, the heart is completely regenerated within 60 days.
In 2015, Panáková had the idea of transferring her team’s years of work on embryonic hearts to the heart of the adult zebrafish. She wanted to focus on the restructuring processes that take place in the heart following a heart attack. These can be methodically stimulated in zebrafish using cryoablation. This technique, which is widely used in zebrafish labs throughout the world, involves injuring a part of the fish’s heart with an ice-cold copper probe. This intervention, which takes no more than five minutes, causes heart cells to die – just as in a heart attack. While the human heart carries scars from this event and is functionally impaired, the zebrafish heart makes a complete recovery. Daniela Panáková hoped that cryoablation would provide vital clues as to why the damaged heart muscle in a zebrafish can recover, so that new treatment concepts could be developed for human heart attack patients.
Working together for animal welfare
We wanted to find out whether administering a painkiller reduced stress in the animals after the intervention and so improved their wellbeing.
Since she was the first Berlin-based researcher to apply to use the method with Berlin’s State Office for Health and Social Affairs (LAGeSo), she got in touch with the MDC’s animal welfare officers and veterinary surgeons. Together, they decided to take account of the most recent assessments of stress in zebrafish when using this intervention. “We wanted to find out whether administering a painkiller reduced stress in the animals after the intervention and so improved their wellbeing,” Panáková explains.
In retrospect, she realises that she and her team took on a notable scientific and communicative challenge. Drawing on all the available expertise, the aim was to come up with improved experiment instructions that would serve as a basis for all future research. Being of the view that animal welfare takes top priority, the team assessed the technical possibilities, the scientific data available on the use of analgesics in zebrafish, and potential effects on the experiment.
The question of pain
In response to pain, mice draw in their whiskers and the nose and cheeks bulge. Animal care staff learn this in advanced training so that they can distinguish between pain and normal states. They notice such changes in behaviour because the signs of pain and stress, such as the pain grimace in mice, are defined. But it was only recently established beyond doubt that zebrafish experience pain and stress. For a long time, the data was based only on insights from the food fish industry. Intensive work over the last five years has resulted in the definition of traits through which zebrafish express stress and pain. However, it is still a challenging task for researchers to recognise and assess them as few laboratories have the methodological prerequisites or expertise to perform detailed experiments in behavioural biology.
For the MDC team, the hard work paid off – and in October 2020, the data was published as a preprint on bioRxiv. First author Sara Lelek recommends morphine to alleviate postoperative pain in zebrafish. Comparative tests also show that morphine does not affect the regeneration of the heart. The team had managed to answer questions relating to animal welfare in the context of biomedical research – a rare combination.
Invitations to international conferences followed. Other scientists, who want to use analgesics to reduce the stress experienced by zebrafish in connection with other methods, have expressed an interest in the data. On the basis of these optimised experiment instructions, Panáková and her colleagues were also able to show which cell types in the injured zebrafish heart play a role and when in order to replace the scarred infarct tissue with newly formed heart muscle cells. For Daniela Panáková, all of this is the best motivation to carry on and make lasting contributions to human and animal health – a mission that makes this researcher’s heart beat.
Text: Anne Merks
Further information
Literature
Sara Lelek et al. (2020): „Morphine alleviates pain after heart cryoinjury in zebrafish without impeding regeneration“. Biorxiv, DOI: 10.1101/2020.10.01.322560.
Bo Hu et al. (2021): „Cellular drivers of injury response and regeneration in the adult zebrafish heart“. Biorxiv, DOI: 10.1101/2021.01.07.425670.