Shedding new light on Leigh syndrome with organoids
Leigh syndrome, also known as subacute necrotizing encephalomyelopathy, is one of the most severe inherited brain diseases in children. In Leigh syndrome, various gene mutations disrupt the mitochondria, which play a key role in maintaining the body’s energy balance. Mutations of the nuclear gene SURF1 are among the ones most often observed. Affected children usually become ill during the first months of their lives, suffering from movement disorders, as well as breathing and swallowing difficulties, and intellectual disabilities. Most children die within a few months or years. Therapies do not exist.
Finding potential targets for therapeutic treatment
In this study, we show that model systems made from patients’ iPS cells can open the way to therapies for a rare disease with a high medical need.
Until now, there has been no effective model for Leigh syndrome caused by SURF1 defects that can help scientists understand the molecular mechanisms of the disease,” explains Professor Alessandro Prigione, who studies stem cell metabolism with his team at the Department of General Pediatrics at the University Hospital of Düsseldorf. In collaboration with research groups led by Professor Nikolaus Rajewsky, Scientific Director of the Berlin Institute for Medical Systems Biology (BIMSB) at the Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC), and Professor Markus Schuelke of the Department of Neuropediatrics at Charité – Universitätsmedizin Berlin, Prigione’s team of scientists has now developed the first organoid model of Leigh syndrome. The model, which they present in the journal “Nature Communications”, is based on induced pluripotent stem (iPS) cells modified with the CRISPR-Cas9 gene-editing tool. “In this study, we show that model systems made from patients’ iPS cells can open the way to therapies for a rare disease with a high medical need,” says lead author Dr. Gizem Inak, summarizing the significance of their work.
Scientists have made many attempts in the past to study mitochondrial diseases in animal models. “But the mitochondrial genome is very different from nuclear DNA,” Prigione explains. “Methods that can modify genes in the nucleus simply don’t work for mitochondria.” Several years ago, Prigione, then a Delbrück Fellow at the MDC, and his research team developed a cell-based model for mitochondrial diseases. The scientists reverted patients’ skin cells to their former state, creating iPS cells. In a further step, they converted these into neurons.
Discovering new uses for old drugs
The scientists found that the progenitor cells of neurons depend on the metabolic processes of mitochondria. As these can be influenced by drugs, the progenitor cells are suitable as a model for drug development, and can be used to test how different substances work. As coordinator of a consortium funded by the European Joint Program on Rare Diseases (EJP-RD), Prigione is using this method to search for therapeutic agents that are already approved for other indications, but which may also be effective against Leigh syndrome. The advantage is that the test phase is much shorter for drugs that have already been tested by the regulatory agencies. Clinical trials can begin sooner, enabling new therapies to be made available more quickly.
Modeling brain development with organoids
Organoids make it possible to model complex human brain development to a certain extent. This is not really possible with standard monolayer cell cultures alone
For the current study, Gizem Inak also created iPS cells from patients’ cells. Using the CRISPR-Cas9 gene-editing tool, she corrected the SURF1 mutation from them. In parallel, she introduced the mutated SURF1 gene into healthy control iPS cells. Then the scientists went one step further. Dr. Agnieszka Rybak-Wolf, who heads MDC’s Organoids Platform, grew organoids from the genetically modified stem cells. “Organoids make it possible to model complex human brain development to a certain extent. This is not really possible with standard monolayer cell cultures alone,” says Rybak-Wolf. Organoids are miniature organs, barely the size of a pinhead, in which cells arrange themselves in three dimensions and mirror some of the structures of the original organ in a petri dish – in this case, the brains of people with and without Leigh syndrome.
In this way, the scientists discovered that the neuronal defects in Leigh syndrome may be caused by an energy deficit at the progenitor cell level. “The patients’ progenitor cells did not form branches, nor did they differentiate into neurons,” Prigione says, describing the stagnation at the neural precursor level. “It was incredible what happened in the petri dish after the mutation was corrected,” says Rybak-Wolf. “The organoid started to develop. In the end, it looked almost exactly like healthy organoids.”
Leigh syndrome is a neurodevelopmental disorder
But that was not the only surprise the scientists uncovered. Leigh syndrome was previously thought to be a neurodegenerative disease triggered by free radical damage to neurons. “By demonstrating that the impaired cell metabolism already affects neural progenitor cells, we have shown that Leigh syndrome is not exclusively a neurodegenerative disease in which neurons form and then die,” summarizes Gizem Inak. “It is more of a neurodevelopmental disorder.” This could also explain why children with Leigh syndrome often have a comparatively small head size and suffer from intellectual disabilities.
Avenues for new treatment strategies
We can use this model to search for the most effective therapeutic strategies against this detrimental diseases affecting children.
“This insight into the underlying mechanism of the disease can now be used to develop targeted treatment strategies for children suffering from this rare disorder,” Prigione says. One way could be to improve the reduced energy output in the progenitor cells using SURF1 gene therapy. This would not involve removing the mutated SURF1 gene, but instead inserting a healthy copy of gene that can work properly, a process known as gene augmentation therapy (GAT). This approach is already being tested for other neurological diseases. In addition, the authors found that one potential candidate for medication therapy is bezafibrate, a drug approved for dietary support in adults. It not only stimulates lipid metabolism, but also activates PGC‑1 alpha, which regulates cell metabolism. “Generating a disease model was the first step” summarizes Prigione, “now we can use this model to search for the most effective therapeutic strategies against this detrimental diseases affecting children”.
Text: Jana Ehrhardt-Joswig
Further information
- A cellular system makes the battle against a rare disease personal
- Alessandro Prigione: Modeling mitochondrial disease
Literature
Gizem Inak et. al: “Defective metabolic programming impairs early neuronal morphogenesis in neural cultures and an organoid model of Leigh Syndrome,” in Nature Communications, DOI: 10.1038/s41467-021 – 22117‑z
Downloads
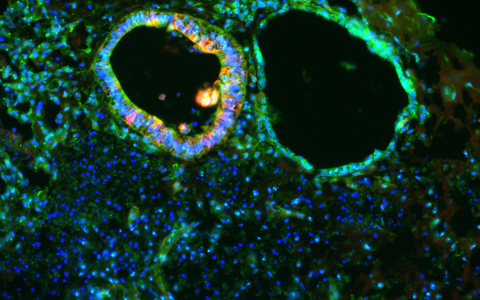
SURF1 organoids show defective generation of neurons (red) and disorganized distribution of neural progenitor cells (green). © Dr. Rybak-Wolf, Organoids Platform, MDC
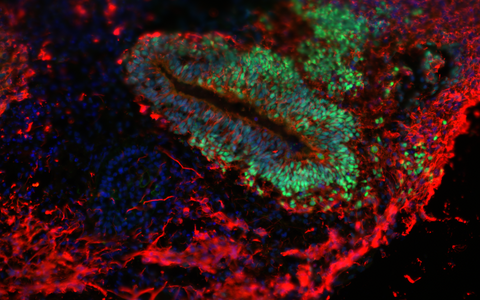
In the organoids in which the SURF1 mutation was genetically corrected, the neurogenic progenitor zones (green) are restored, and mature neurons (red) can be generated. © Dr. Rybak-Wolf, Organoids Platform, MDC
Contacts
Professor Alessandro Prigione
Düsseldorf University Hospital
Clinic for General Pediatrics, Neonatology and Pediatric Cardiology
Tel.: +49 – 211-8118705
alessandro.prigione@hhu.de
Jana Ehrhardt-Joswig
Editor, Communications Department
Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC)
Tel.: +49 – 30-9406 – 2118
jana.ehrhardt-joswig@mdc-berlin.de or presse@mdc-berlin.de