Towards a better understanding of self-healing
Embryonic cells can change into many different cell types to form different organs. In adult cells, however, this ability is gradually lost: once mature, their fate is largely already decided. This is why the ability to self-heal in humans is limited.
In many other species, this isn’t the case. “In zebrafish, vital organs like the heart, brain and pancreas are capable of regenerating and regaining their normal functions even after serious injury,” explains Dr Jan Philipp Junker, who leads the Quantitative Developmental Biology lab at the Berlin Institute for Medical Systems Biology (BIMSB), part of the Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC). “To do this, the organs use very different mechanisms,” he adds.
Understanding the zebrafish’s magic trick
The systems biologist wants to find out how zebrafish achieve this regeneration, with a view to potentially developing better treatments for people who have diabetes or have had a heart attack or stroke, for example. For his current research project, planned together with developmental biologist Professor Nikolay Ninov from the Center for Regenerative Therapies Dresden (CRTD) at TU Dresden, the two researchers have received a DFG Sequencing Grant.
The purpose of these grants is to support important projects that use next-generation sequencing (NGS). This term covers a range of modern and usually very expensive sequencing techniques. “I’m very pleased that, with the help of the grant, we can now go ahead with this ambitious project over the next three years,” says Junker. The DFG will fund a doctoral and a postdoctoral position and consumable materials as well as providing the two researchers with a budget of around €380,000 for the sequencing work.
Watching genes at work
Together with their teams, Junker and Ninov intend to take a closer look at islet cells in the pancreas of zebrafish. One group of these cells, known as beta cells, is responsible for making insulin, which controls blood sugar levels. If the beta cells die due to injury, other islet cells – alpha and delta cells – will start to transform and produce insulin too. This means that zebrafish always have a means to regulate blood sugar. Human islet cells lack this ability – which is why people whose beta cells are destroyed by the immune system develop type 1 diabetes.
“We want to find out how the fate of a cell is decided during embryonic development and regeneration, and whether both involve similar molecular processes,” says Junker. “We’ll be using a new method to determine which genes are active in individual cells at any one time and also which genes were expressed several hours before.” This will make it possible to observe more accurately how islet cells change from one type to another.
Removing barriers
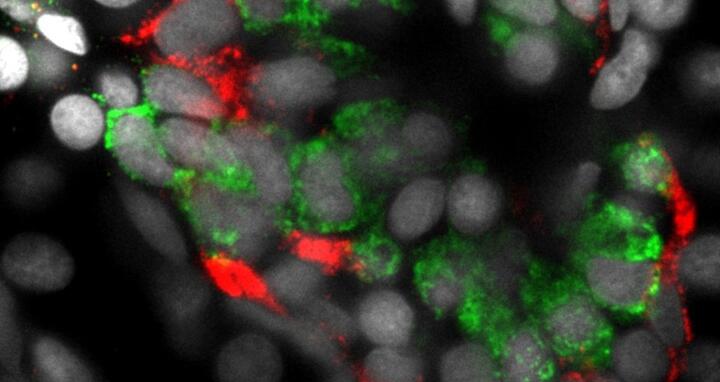
This image shows islet cells in the pancreas of a zebrafish. The insulin-producing beta cells are shown in green, and the glucagon-producing alpha cells are shown in red.
To understand the mechanisms that control the activation and deactivation of genes, the researchers also plan to investigate which genome segments are accessible for reading the genes and which are blocked by proteins. “You can think of it as being like a roadblock,” says Junker. “Once we discover the barriers that prevent a cell from transforming, we can assume that it will be possible to remove them – and therefore switch the genes involved in the transformation on and off as required.”
Finally, the team plan to compare the zebrafish islet cells with human islet cells. “It’s possible that the molecular barriers are fairly similar to those in zebrafish,” Junker speculates. The vision, at least, is there: with the help of suitable techniques, one day it might be possible to simply tear down the roadblocks that prevent alpha and delta cells from changing into insulin-producing cells. We would then have a cure for type 1 diabetes.
Text: Anke Brodmerkel