Simon Lab
Cell Biology of Immunity
Profile
Cells are specialists in sustainability. They eat up all the organelles or larger molecular complexes that they no longer need – in order to then recycle the digestion products to build new structures. Autophagy is the name of this cellular recycling process, which yeast, but also all plant, animal and human cells use for themselves. Without autophagy, cells cannot develop and function normally, they age faster without it, and so does the immune system. In our lab we investigate the role of autophagy in the immune system and how to use autophagy to keep the immune system young.
Team
Group Leader
Prof. Dr. Katja Simon
- Curriculum Vitae (CV)
2021-present W3 Professor at Max Delbrück Center for Molecular Medicine, Berlin,
Distinguished Professor of Helmholtz Association Award
2016 – 2026 Senior Group Leader at Kennedy Institute of Rheumatology, Oxford
Wellcome Trust Senior Investigator Award, 2015 – 2020 and 2022 – 2027
2015 Professor of Immunology, Oxford 2014 Associate Professor, Oxford 2008 – 2015 Head of the Translational Immunology Laboratory, Oxford 2006 University Research Lecturer, Oxford 2004-present Principal Investigator with independent grant funding, Weatherall Institute of Molecular Medicine, Oxford 1998 – 2004 Postdoc, Weatherall Institute of Molecular Medicine, Oxford (Gavin Screaton) 1994 – 1998 Postdoc, Centre d’Immunologie Marseille (Anne Marie Schmitt-Verhulst) 1994 First PhD student of Deutsches Rheumaforschungszentrum (Avrion Mitchison) 1990 Diploma in Biology at Freie Universität Berlin & Master Immunology UCL London
Administrative Assistant
Daniela Keyner
Contact: dkeyner@mdc-berlin.de | +49 30 9406 – 3726
Merit Seeliger
Contact: Merit.Seeliger@mdc-berlin.de
Scientists
Dr. Elisabeth Greßler
Contact: AnneElisabeth.Gressler@mdc-berlin.de | +49 30 9406 – 3232
Contact: Sebastian.Hofer@mdc-berlin.de | +49 30 9406 – 2372
Dr. Timo Benedikt Trefzer
Contact: TimoBenedikt.Trefzer@mdc-berlin.de
Timo is a computational biologist focused on understanding how the immune system changes with age. He leverages cutting-edge omics technologies and integrative multi-omics approaches to uncover the complex molecular mechanisms that drive immune aging. He earned his PhD developing and applying single-cell RNA sequencing (scRNA-seq) technologies, using them to study lung cancer in young female never-smokers. Through his work, Timo combines technological innovation with biological insight to address pressing questions in human health and disease.
PhD Students
Jakub Belhadj
Contact: Jakub.Belhadj@mdc-berlin.de
Jakub is a PhD student funded by UNION, an EU MSCA Doctoral Network. His research focuses on characterizing how mitophagy is altered during aging, evaluating whether these changes are detrimental, and exploring how restoring mitophagy can rejuvenate immune cell function to prolong healthspan. Jakub earned his BSc and MSc from the University of Wrocław and the Polish Academy of Sciences. Prior to joining the lab, he completed an internship at VIB-KU Leuven and worked as a Research Assistant for three years at the Medical University of Vienna.
Matisse da Silveira
Contact: matisse.dasilveira@mdc-berlin.de
Matisse is a PhD student interested in the impact of fasting on vaccine responses and immune resilience in aged organisms. He aims to explore the underlying molecular mechanism with the help of flow cytometry, proteomics, and metabolomics. Training as a chemist and biologist, Matisse obtained a Engineering degree (Msc) from the University of Strasbourg, and broadened his horizon with studies and lab internships at Alberta University, Canada and at Merck healthcare, Switzerland. His PhD is funded by UNION, an EU MSCA Doctoral Network.
Anushree Gaigore
Contact: Anushree.Gaigore@mdc-berlin.de
Anushree joined the Simon Lab in 2024 as a PhD student and is funded by the SFB 1177 Molecular and Functional Characterization of Selective Autophagy. Her research focuses on understanding how autophagy controls bone marrow adiposity, inflammation and aging. She completed her M.Sc. in Biotechnology from Symbiosis School of Biological Sciences, Pune, and subsequently gained research experience at the Indian Institute of Science, Bangalore, and Duke-NUS Medical School, Singapore
Aikaterini Giannopoulou
Contact: Aikaterini.Giannopoulou@mdc-berlin.de | +49 30 9406 – 3233
Katia is a PhD student from Greece investigating the immunological and metabolic effects of amino acid restriction in healthy individuals. Her research seeks to bridge systemic metabolic shifts with cellular-level changes including autophagy, in immune populations using flow cytometry. By integrating proteomic and metabolomic analyses, Katia aims to uncover the molecular mechanisms underlying amino acid restriction, ultimately offering new insights into how dietary interventions influence the human immune system and promote healthy aging.
Simon Christof Rapp
Contact: SimonChristof.Rapp@mdc-berlin.de
Simon is a PhD student interested in the impact aging on the function of lysosomes in human T cells. He aims to explore their underlying molecular profile with the help of flow cytometry, proteomics, and metabolomics. Training as a biochemist, Simon obtained his BSc and MSc at Heidelberg University, and broadened his horizon with studies and lab internships in London, Oxford, and Berkeley
Carolin Vats
Contact: Carolin.Knappe@mdc-berlin.de
Carolin investigates how tissues shape mitochondrial quality control in long-lived macrophages and how this determines their function during ageing. By combining mitochondrial genetics, mitophagy reporters, and metabolic profiling, she explores how local microenvironments drive mitochondrial selection — with the goal of preserving immune resilience and tissue health across the lifespan.
Technicians
Sandra Neuendorf
Contact: sneuen@mdc-berlin.de | +49 30 9406 – 3234
Sandra earned her diploma degree in Biotechnology from Beuth University of Applied Sciences in Berlin. Joining MDC in 2007, she worked with Prof Wanker, focusing on neurodegeneration and neuroproteomics, before moving to Dr Zsuzsanna Izsvák’s group to work on the Sleeping Beauty technology. Since September 2025, she has been a member of Prof. Katja Simon’s lab, shifting her focus to the immune system. Sandra manages and organizes ongoing clinical trials at the intersection of nutrition and autophagy. She values working in diverse and international teams and is committed to supporting the next generation of researchers.
Isabel Panse
Contact: Isabel.Panse@mdc-berlin.de | +49 30 9406 – 3234
Isabel is a biological technical assistant from Berlin with a strong research background in Immunology. After working for many years at the Charité and the DRFZ, she joined Katja’s research group in Oxford in 2014, where she investigated the role of autophagy in human immune cells and provided essential technical and organizational support. In 2022, she helped Katja to relocate her lab to Berlin. Since then, Isabel has been working at the MDC as a lab manager, supporting Katja’s team with administrative responsibilities as well as laboratory work.
Ramona Zummach
Contact: r.zummach@mdc-berlin.de | +49 30 9406 – 3234
Ramona completed her training as a chemical laboratory assistant at the Central Institute for Molecular Biology of the Academy of Sciences of the GDR before pursuing a degree in chemical engineering with a focus on environmental analysis at the State Technical School in Berlin. For many years, she worked as a technical assistant in Dr. Schunck’s “Cytochrome P450 and ER” research group. She joined the Simon lab in April 2022. She is passionate about laboratory work and values the diverse challenges that research presents.
Graduate & Undergraduate Students
Jana Hadatsch
For an internship and her masters project, Jana is studying the function of lysosomes and their link to STING signaling in young and old. To investigate this, she is using CRISPR/Cas9 genetic perturbation as well as pharmacological interventions.
Klaudia Milaj
Klaudia is a master’s student from Greece investigating the physiology of macrophages in the presence of pathogens. Using pharmacological interventions, she aims to understand which pathways are involved in macrophage phagocytosis.
Trainee
Ellen Kathleen Trombalski
Research
Macroautophagy is the major degradation pathway in the cell responsible for the turnover of bulk cytoplasmic components. In contrast to the ubiquitin – proteasome system, which primarily degrades individual proteins, macroautophagy sequesters and degrades entire organelles — including mitochondria, lipid droplets, ribosomes, and endoplasmic reticulum, recycling their building blocks. Macroautophagy thereby plays a central role in regulating cellular metabolism and key cell fate decisions, including apoptosis, differentiation, and cell cycle progression.
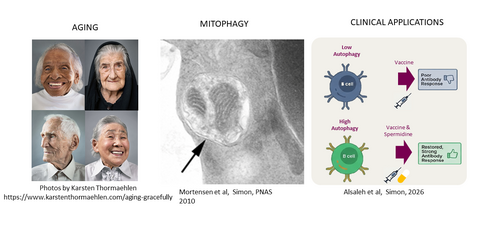
Our group has been at the forefront of elucidating the role of autophagy in immune cell development, function, and metabolism. Using tissue-specific in vivo models, we demonstrated that autophagy is essential for red blood cell maturation through mitochondrial clearance (Mortensen et al, PNAS 2010). We further established that autophagy drives neutrophil differentiation by supplying free fatty acids, highlighting its metabolic importance in immune cell development (Riffelmacher et al, Immunity 2017).
We were the first to show that autophagy is indispensable for stem cell maintenance, as its absence leads to the loss of hematopoietic stem cells (Mortensen, J Exp Med et al, 2011). Extending these findings, we identified a critical role for autophagy in the maintenance of B1 cells, a self-renewing B cell population (Clarke et al, JEM 2018). Similarly, we uncovered that memory T cells — characterized by self-renewal and stem-like properties — depend on autophagy. Notably, during the first division following antigen encounter, memory T cells undergo asymmetric mitochondrial inheritance, a process further reinforced by autophagy to support asymmetric cell fate decisions (Borsa et al, Nat Cell Biol 2025).
We discovered that the endogenous metabolite spermidine is required for autophagy in memory T amd B cells and can be supplemented through dietary means when endogenous levels decline (Puleston et al, elife 2014). Mechanistically, we defined spermidine’s mode of action via hypusination of eIF5A (Zhang et al, Mol Cell 2019). In parallel, we developed pioneering methods to measure autophagy in primary cells, revealing a decline in both autophagy in aging T lymphocytes and also found spermidine levels reduced (Phadwal et al, Autophagy 2012, Alsaleh et al, elife 2020). Collectively, these findings demonstrate that autophagy is a critical regulator of immune system aging.
Given the central role of memory T cells in vaccine responses, our work suggested that enhancing autophagy could improve vaccine efficacy in older individuals. This hypothesis has been validated in a double-blind, placebo-controlled clinical trial demonstrating that spermidine supplementation enhances autophagy and improves B cell responses to vaccination in older adults (NCT05421546).
Building on these discoveries, we are now investigating the autophagosomal cargo to understand autophagy’s role in immune cells more mechanistically. We developed a novel mouse model enabling proximity labeling of autophagosomal contents, leading to the identification of a key cytokine receptor that explains impaired CD4⁺ T cell proliferation in autophagy-deficient cells (Zhou et al, Nat Comms 2022). More recently, we have expanded our focus to cell-extrinsic roles of autophagy, demonstrating that autophagy in adipocytes contributes to intercellular signaling that modulates macrophage function (Richter et al, EMBO 2023, Piletic et al, Nat Comms 2025).
Together, our research establishes autophagy as a central regulator of immune cell biology across cell intrinsic and extrinsic contexts. Leveraging this foundation, we aim to identify novel therapeutic targets for regenerative medicine and vaccination in older adults, as well as develop robust biomarkers to monitor autophagy in clinical settings.