Ancient viral DNA drives preeclampsia in pregnancy
Preeclampsia is a dangerous pregnancy complication that arises from abnormal placental development, which can threaten both mother and baby.
In a study published in “Genome Biology,” researchers from the Max Delbrück Center, Institut Necker Enfants and colleagues from the University of Bath show that fragments of ancient viral DNA embedded in our genomes act as essential control switches on placental genes. Their central finding: an endogenous retroviral element called LTR8B is required for expression of the Pregnancy Specific Glycoprotein (PSG) gene cluster, including PSG9 – a gene strongly associated with preeclampsia.
“This work shows that an ancient viral sequence can act like a manual for a placental gene,” explains Dr. Manvendra Singh, a lead author of the study and former postdoc in the Mobile DNA Laboratory led by Professor Zsuzsanna Izsvák at the Max Delbrück Center, and who is now Group Leader at the Institut Necker Enfants and Institut Imagine. “By mapping the regulatory region and testing it functionally, we have now connected genome evolution to a concrete disease mechanism – and to the possibility of earlier detection.”
Multiple layers of control
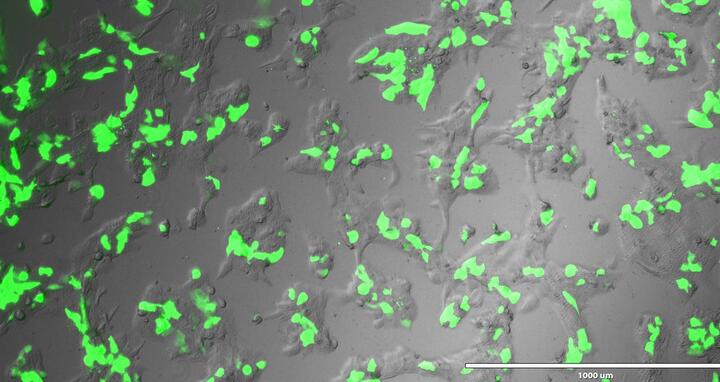
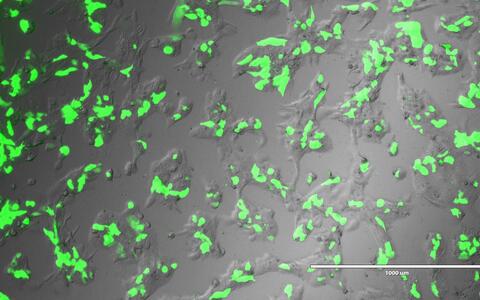
Human placental cell expressing Green Florescent Protein, which is used as a marker to identify cells that have successfully taken up an introduced gene sequence.
Endogenous retroviruses are viral sequences left behind by infections that occurred millions of years ago. Most are usually silenced, but some have been co-opted by evolution. It is now generally now accepted that the placenta evolved by co-opting retroviral genes. These endogenous retroviral sequences that are now part of our genome regulate nearby genes, and also reorganize placental gene expression.
By editing PSG genes in placental cell models, the team discovered that the viral regulatory element LTR8B plays a key role in placental development. LTR8B is found repeatedly in the PSG (PSG1-11) gene cluster and orchestrates its expression by recruiting key placental transcription factors. When a single repeat was removed, expression of the PSG gene cluster collapsed, which disrupted placental development.
The team also found that a second viral element, MER65, is involved in regulating PSG genes. MER65 helps determine which forms of PSG proteins are released from cells and circulate in the mother’s bloodstream, where they can be measured in blood tests. Together, LTR8B and MER65 present examples of viral elements helping to determine when a gene is turned on, how it is processed, and where the resulting product ends up.
“What made this discovery powerful was being able to move from prediction to direct functional tests,” says Izsvák, who led the experimental work at the Max Delbrück Center and is a corresponding author on the paper. “The placenta depends on precisely tuned gene regulation, and retroviral elements sit at the center of that tuning.”
Why it matters for preeclampsia
“Preeclampsia is both remarkably common (about 5% of pregnancies) and potentially lethal,” says Professor Laurence Hurst at the University of Bath’s Milner Centre for Evolution, a co-corresponding author. “It is the second most common killer of mothers globally, making it both an evolutionary conundrum and a serious maternal and neonate health problem. Unfortunately, preeclampsia is often recognized too late – when a pregnant woman’s blood pressure is already dangerously very high.”
“Since expression of the PSG9 gene is higher than normal in preeclampsia, we now have a realistic prospect of improving early diagnosis, before the onset of symptoms,” adds Hurst.
The team, in collaboration with Professor Sandra Blois at the University Medical Center Hamburg-Eppendorf, is now working to validate PSG9 as a clinical biomarker in larger pregnancy cohorts. More broadly, they aim to test whether retroviral regulatory circuits can be used to stratify risk or to reveal therapeutic entry points in placental disorders.
The research is a follow-up of the work published last year by the same team, who reported on how virus-derived regulatory elements, integrated into the process of placental development during evolution, are responsible for the abnormal expression of multiple genes and this in turn contributes to preeclampsia during pregnancy.
Text: Manvendra Singh, Gunjan Sinha
Further information
Ancient viral DNA shapes modern human placentas
Literature
Manvendra Singh, Yuliang Qu, Amit Pande, et. al. (2026) “Endogenous retroviral elements LTR8B and MER65 rewire PSG9 regulation to control trophoblast syncytialization and pre-eclampsia risk” Genome Biology DOI: 10.1186/s13059-026 – 03944‑z
Contacts
Dr. Manvendra Singh
Group Leader
Systems Biology of Mobile DNA and Genome Regulation
Institut Necker Enfants Malades
Institut Imagine
manvendra.singh@inserm.fr
Lama Awada
Press Office
Institut Imagine
lama.awada@institutimagine.org
+33 6 35 12 02 61
Corinne de Conti
Communication and Public Affairs Director
Institut Imagine
corinne.deconti@institutimagine.org
+33 6 72 38 76 42
Vicky Just
Media and PR Manager
University of Bath, UK
+44 (0)1225 386883
vjj21@bath.ac.uk or press@bath.ac.uk
Gunjan Sinha
Editor, Communications
Max Delbrück Center
+49 30 9406 – 2118
Gunjan.Sinha@mdc-berlin.de or presse@mdc-berlin.de
- Max Delbrück Center
-
The Max Delbrück Center for Molecular Medicine in the Helmholtz Association lays the foundation for the medicine of tomorrow through our discoveries of today. At locations in Berlin-Buch, Berlin-Mitte, Heidelberg, and Mannheim, interdisciplinary teams investigate the complexity of disease at the systems level – from molecules and cells to organs and entire organisms. Together with academic, clinical, and industry partners, and as part of global networks, we turn biological insights into innovations for early detection, personalized therapies, and disease prevention. Founded in 1992, the Max Delbrück Center is home to a vibrant, international research community of around 1,800 people from over 70 countries. We are 90 percent funded by the German federal government and 10 percent by the state of Berlin.