How blood and leukemia cells develop
Scientists from the Berlin Institute of Health at Charité (BIH) and the Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC), together with colleagues from Heidelberg Institute of Stem Cell Research and Experimental Medicine (HI-STEM) at the German Cancer Research Center (DKFZ) and more researchers from Heidelberg and Barcelona, have traced the development of blood cells in detail: They combined methods used to analyze gene activity within the cell with those used to detect protein molecules on the cell surface. This enabled them to simultaneously capture information on thousands of individual cells from blood and bone marrow and thus clearly identify the different developmental stages of the various cell types. The scientists have now published their findings, which are important for the diagnosis and treatment of blood cancers, in the journal Nature Immunology.
Detect subtle differences
To the layman, blood cells consist of red blood cells, white blood cells and platelets. Experts, however, make many more distinctions. This is especially true in the case of white blood cells – the immune cells – which are composed of many different cell types. What all of these cells have in common is that they originate from blood stem cells in the bone marrow. Blood cell diversity arises when these blood stem cells divide and differentiate into different cells. “Up until now we have used the proteins on the cell surface to distinguish between the different cells,” explains Dr. Simon Haas, co-last author of the current paper and head of the Junior Research Group “Systems Hematology, Stem Cells & Precision Medicine” at the BIH, which is based at the Berlin Institute for Molecular Systems Biology (BIMSB) of the MDC. “We do this by employing FACS analysis, a method based on flow cytometry, which involves analyzing cells with the help of antibodies against about 20 surface molecules,” explains Haas. “It’s fast and inexpensive, but it’s not that precise.”
Single-cell analysis allows us to detect much finer differences between cells.
In the meantime, there are much more precise methods for examining cells. Bioscientists can now use single-cell analysis to determine the various messenger RNA (mRNA) molecules in individual cells. These messenger molecules relay information from the genome in the cell nucleus – the DNA – to the cytoplasm, where the mRNA is translated into protein. The amount and composition of mRNA may vary in each cell. “This allows us to discern much finer differences between cells,” says Haas. “As a result, we can identify different stages of cell development, which is important when it comes to such things as more accurately diagnosing blood cancer and understanding the aging process of blood cells.” Yet single-cell diagnostics is still far too time-consuming and expensive for daily clinical use. “So we had the idea to combine single-cell analysis with FACS analysis,” recounts Haas.
Highly accurate map of blood development
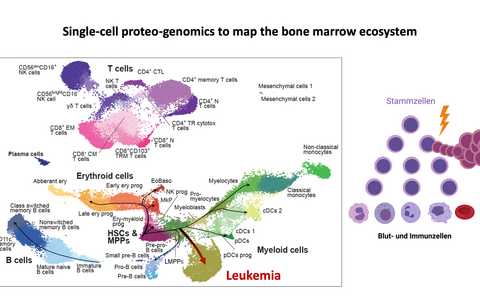
To this end, the scientists led by Simon Haas, together with colleagues at the Heidelberg Institute for Stem Cell Technology and Experimental Medicine (HI-STEM), the German Cancer Research Center (DKFZ), the European Molecular Biology Laboratory (EMBL) in Heidelberg and the Centre for Genomic Regulation (CRG) in Barcelona, first examined hundreds of thousands of blood cells at different stages in the laboratory using single-cell analysis and thus created a highly accurate map of the development of blood system cells, both from healthy donors and leukemia patients. This enabled them to understand the normal aging processes, but also to see how stem cells give rise to leukemia cells. During their investigations, they also discovered new marker proteins on the cell surfaces that allow for much more precise distinctions between the different cell types.
“This is very important in the diagnosis and treatment of blood cancers,” stresses Haas, “where the cells do not develop into functionally complete blood cells, but stop at a certain stage during their development and keep multiplying. This continues until the blood is flooded with white blood cells, the hallmark of leukemia, which literally means ‘white blood.’ In order for us to target these cells, it is necessary to have precise knowledge of them and be able to distinguish them from healthy blood cells.”
Combining analytical methods for precision diagnostics
The scientists linked the newly discovered information about the different cell stages with the conventional surface markers used in FACS. Incorporating the data about the new marker proteins, they were able to use artificial intelligence (AI) to combine the results from the single-cell analyses with the FACS results. “We now use new combinations of antibodies suggested by AI to better identify very specific cell stages,” explains Haas.
Haas’s junior research group is part of the joint research focus “Single-Cell Approaches to Personalized Medicine,” which the BIH at Charité launched together with the MDC and Charité – Universitätsmedizin Berlin. Haas is now pursuing the clinical application of his findings together with his clinical partners, Professor Lars Bullinger and Professor Ulrich Keller, the Directors of the Department of Hematology, Oncology and Tumor Immunology at Charité Campus Virchow-Klinikum (CVK) and Charité Campus Benjamin Franklin (CBF), respectively. “Our aim is to improve the diagnosis of leukemias and to be able to fight blood cancer cells in a targeted way.”
Text: BIH
Further information
- Single-cell analysis at the MDC
- Focus area “Single cell approaches for personalized medicine”
- Berlin Cell Hospital
Literature
Sergio H. Triana et al. (2021): “Single-cell proteo-genomic reference maps of the hematopoietic system enable the purification and massive profiling of precisely defined cell states”. Nature Immunology, DOI: 10.1038/s41590-021 – 01059‑0
- Berlin Institute of Health in der Charité (BIH)
-
The mission of the Berlin Institute of Health (BIH) is medical translation: transferring biomedical research findings into novel approaches to personalized prediction, prevention, diagnostics and therapies and, conversely, using clinical observations to develop new research ideas. The aim is to deliver relevant medical benefits to patients and the population at large. As the translational research unit within Charité, the BIH is also committed to establishing a comprehensive translational ecosystem – one that places emphasis on a system-wide understanding of health and disease and that promotes change in the biomedical translational research culture. The BIH was founded in 2013 and is funded 90 percent by the Federal Ministry of Education and Research (BMBF) and 10 percent by the State of Berlin. The founding institutions, Charité – Universitätsmedizin Berlin and Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC), were independent, member entities within the BIH until 2020. Since 2021 the BIH has been integrated into Charité as the so-called third pillar. The MDC is now the Privileged Partner of the BIH.
- Max Delbrück Center for Molecular Medicine (MDC)
-
The Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC) is one of the world’s leading biomedical research institutions. Max Delbrück, a Berlin native, was a Nobel laureate and one of the founders of molecular biology. At the MDC’s locations in Berlin-Buch and Mitte, researchers from some 60 countries analyze the human system – investigating the biological foundations of life from its most elementary building blocks to systems-wide mechanisms. By understanding what regulates or disrupts the dynamic equilibrium in a cell, an organ, or the entire body, we can prevent diseases, diagnose them earlier, and stop their progression with tailored therapies. Patients should benefit as soon as possible from basic research discoveries. The MDC therefore supports spin-off creation and participates in collaborative networks. It works in close partnership with Charité – Universitätsmedizin Berlin in the jointly run Experimental and Clinical Research Center (ECRC ), the Berlin Institute of Health (BIH) at Charité, and the German Center for Cardiovascular Research (DZHK). Founded in 1992, the MDC today employs 1,600 people and is funded 90 percent by the German federal government and 10 percent by the State of Berlin. www.mdc-berlin.de