How cells devour themselves
“Sascha Martens and I have always been a super team,” says Oliver Daumke, beaming. He fondly recalls the time when the two men studied together, when they shared a lab bench while doing their thesis, and when they worked side by side as postdocs in the same lab in Cambridge. “We’ve always collaborated well together,” adds Daumke.
That’s still true today, even though the two friends now work in different European cities that are about an eight-hour drive from one another. Whereas Daumke moved from England to Berlin in 2007 to work as a structural biologist at the MDC, Martens ended up at the University of Vienna, where he is investigating cellular autophagy (a Greek term for “self-eating”). For when cells want to dispose of their own intracellular components and recycle them into valuable nutrients, they digest these components in a complex process that is not yet fully understood.
Where cells start munching
How do cells know precisely where they should begin to chomp away? This was a mystery for a long time.
“How do cells know precisely where they should begin to chomp away? This was a mystery for a long time,” says Daumke. In collaboration with James Hurley from the University of California, Berkeley, the scientists have now solved this mystery and are presenting their findings in the journal Molecular Cell.
“Autophagy is a fundamental biological process,” says Daumke. If it doesn’t function properly, it can cause diseases. Damaging deposits form in the nervous system, leading to dementia. Cancer cells use autophagy, on the other hand, as a quick way to get energy and raw materials for their destructive growth. “By having an in-depth understanding of autophagy, we’ll be able to regulate it,” says Daumke – and this may pave the way for treatment of disease.
Numerous proteins help the process along
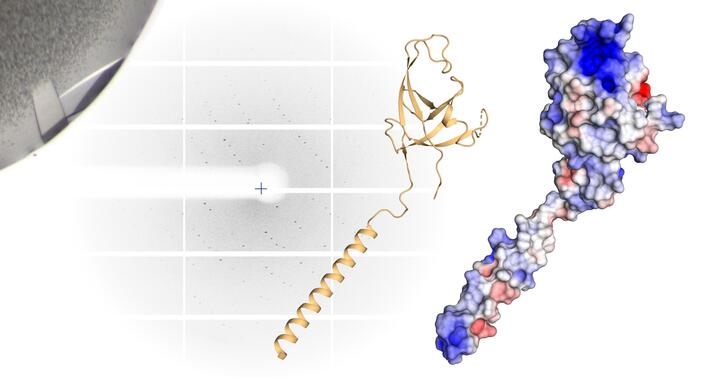
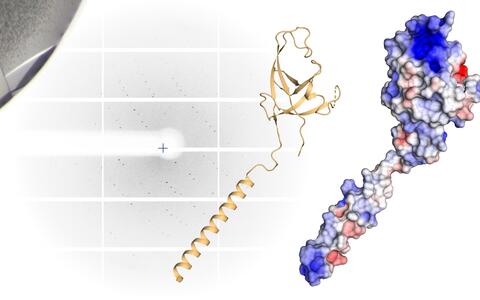
Important for autophagy: In a simplified three-dimensional representation of the "claw", the arm and hand are clearly visible (left). In the room-filling molecule model (right), positive charges are red, negative charges blue - the highly charged spot at the tip of the molecule stands out particularly.
This process brings together myriad molecules in the cell’s cytoplasm. Molecules like the p62 protein lie down on defective, clumped-up proteins, thus making them irresistible to the cell. The cell then puts a membrane around its “food,” much as a human mouth would. Next, digestive enzymes “chew up” everything inside the membrane.
For years Martens had been racking his brain over what exactly happens in the first stage of autophagy – that is, what whets a cell’s appetite for itself. “Whenever we talked on the phone or met, we would discuss this question,” says Daumke. “Sascha never stopped searching for answers.”
One day Martens’s team found signs that the p62 protein recognizes the proteins which need to be digested on the one hand, but on the other comes in direct contact with the cell’s chewing apparatus and thus acts as an adapter. But they lacked the equipment needed to conduct more precise tests. “Sascha wanted to know what occurs at the molecular level – and so he contacted me,” says Daumke.
A molecular claw
Daumke and his team are specialists in the smallest of the small and can even peer into the atoms of a molecule. In the lab, they produce large quantities of proteins, from which they grow crystals, and then bombard these with an intense X-ray beam. The beam is refracted by the crystal and researchers can use the unique arrangement of the diffraction spots to calculate how the proteins are assembled. This creates detailed models that have greater accuracy than a light microscope could ever offer.
Marie Witt, Oliver Daumke and Tobias Bock-Bierbaum
PhD student Marie Witt and postdoctoral researcher Tobias Bock-Bierbaum, both members of Daumke’s lab, took a closer look at the p62 molecule and the components of the autophagy apparatus. They discovered an unusual structure at one end of the FIP200 protein, one that is essential for autophagy. “We found a sort of arm, hand, and finger-like extensions and christened this whole formation the ‘claw,’” says Witt, adding that the inside of the claw has a strong positive electrostatic charge, “and that something like this always raises a flag in the minds of structural biologists.” This is because positively and negatively charged protein building blocks attract one another. “We asked ourselves: ‘Is this perhaps where p62 binds?’”
A strong grip that binds
Daumke then discussed the findings with his friend from university. Further tests in Vienna and Berlin corroborated the scientists’ suspicions: “The FIP200 claw grabs the p62 and doesn’t let it go,” says Bock-Bierbaum. The scientists introduced targeted mutations in order to change the hand’s electrostatic charge. The strong grip loosened. “We are confident we have identified the binding site,” says Witt.
Martens’s team then showed that p62 and the FIP200 claw are also found together in living human cells. When Martens removed the claw from the FIP200 protein, the cell’s appetite for proteins tagged as tasty morsels by p62 diminished, and autophagy slowed down.
Open issues
““We thus proved a link between the cargo that should be digested and the autophagy machinery,” says Witt. “The discovery of this link is very important for autophagy research,” adds Daumke. “We can now reconstruct which components become active and when. That’s a really nice signaling cascade.”
For me, science has always meant doing what you like to do, and working with people you like to work with.
But there are still some unanswered questions. “We would like to know the structure of the complex between p62 and FIP200,” says Daumke. In their experiments, they observed that FIP200 is very weakly bound to its adapter. This, according to Daumke, makes the matter interesting but also more difficult to study.
That also means there are plenty of opportunities for further collaboration with Sascha Martens’s lab in Vienna. But, according to Daumke, there is no lack of those anyway. “Because of our long friendship, we simply do what we enjoy doing and investigate what interests us,” says Daumke. “For me, science has always meant doing what you like to do, and working with people you like to work with.”
Text: Martin Ballaschk
Literature
Eleonora Turco et al. (2019): „FIP200 claw domain binding to p62 promotes autophagosome formation at ubiquitin condensates“. Molecular Cell, Vol 74, 2. DOI:10.1016/j.molcel.2019.01.035