The immune system as a sculptor
The immune system is mind-bogglingly complex and fantastic at the same time
The immune system is more than just a few liters of blood and lymph. It is, in the words of MDC group leader Professor Michela Di Virgilio, “mind-bogglingly complex and fantastic at the same time.” The researcher finds it incredible to observe how an assembly of organs, tissues, and countless types of cells, molecules and genes can work together so flawlessly. “It’s all about balance,” she explains. After all, the body needs to ensure it directs its attacks towards pathogenic invaders and does not turn on itself.
What’s more, it would appear the body’s defense system is also extremely inventive. White blood cells, for example, work like sculptors to forge diverse antibody molecules out of a single DNA template. In her new publication, which appeared in the scientific journal Molecular Cell, Di Virgilio shed lights on one of the mechanisms that guide the sculptor’s hand to the right place.
The antibody sketch, which Michela Di Virgilio has been using to explain how ZMYND8 works during the interview
How cells reassemble antibodies
The scientist sketches an antibody on the back of an old publication. The drawing looks like the letter Y, with one base and two arms stretching upwards to the left and right. This protein swims through all body fluids in innumerable variations, she explains, and uses its “arms” to attach itself to molecules from bacteria, viruses, and other foreign particles. The immune system creates these arm-like “variable regions” using targeted breaks and changes them with mutations, which enable it to recognize the vast variety of different pathogens. As soon as the body’s defenses detect an intruder, the immune system starts to refine its response. All of this happens in the B cells – white blood cells that mature in the bone marrow.
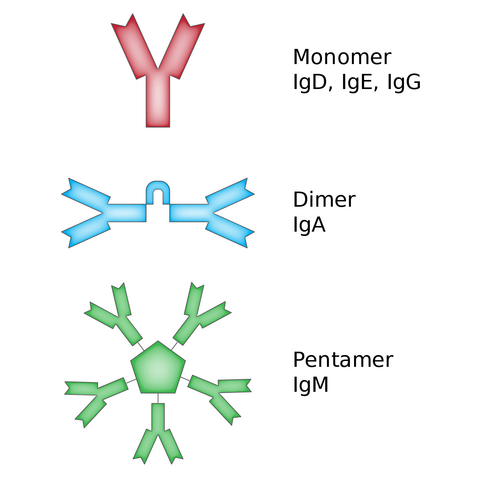
“This part here determines the effector function,” says the researcher, circling the base of the molecule in her sketch. This so-called “constant region” determines the mechanism by which the immune system gets rid of the pathogen. The cell chooses from five different base modules, thus using a single template to create antibody molecules with different functions.
The effector function of antibodies (immunoglobulins, Ig) is determined by the constant region at the “base” of the molecule. Depending on the type or “class” of antibody the proteins appear as single molecules or as assemblies.
In order to attach the “arms” of the antibodies to a new base, enzymes have to break up the DNA and patch it back together again. This a delicate matter, says Di Virgilio, since as both strands of the double helix break, the lesion is extremely difficult to repair. What’s more, if wrong parts of the genome are joined together, cancer can develop. “There is a balance between the formation of DNA breaks on the one hand and their accurate repair on the other.” Exactly how the cells repair these intentionally generated breaks remains an area of intense investigation. “At first, I was mainly interested in the repair process,” says Di Virgilio.
Make and break
In a large-scale experiment, Di Virgilio’s team fished the proteins from the cell that are active during the swapping out of the “base” regions. Among them was a protein called ZMYND8, which binds to DNA and can read information.
Many long evenings in the laboratory ensued, particularly for doctoral student Verónica Delgado-Benito, as the team searched for the repair enzymes that worked together with ZMYND8. “We researched for a role of ZMYND8 in repair for a really long time,” says Di Virgilio. “At some point, we had to admit to ourselves that it had nothing to do with the repair process.” It was at this point that the project took a turn.
The protein, it transpires, is actually involved in breaking apart the DNA, not patching it up. The research team discovered this by using B lymphocytes from the spleen of mice – one of the lymphoid organs in which B cells encounter the pathogen molecules and further mature. Di Virgilio explains that ZMYND8 contributes to guide an extremely aggressive, and even dangerous, enzyme called Activation Induced Deaminase (AID) to precisely the right place. Once there, it initiates the formation of breaks, which results in the DNA thread being smashed like a sledgehammer. “If this enzyme were to strike anywhere else in the genome, the results would be very bad.” But the immune system manages to guide the hammer to just the right spot.
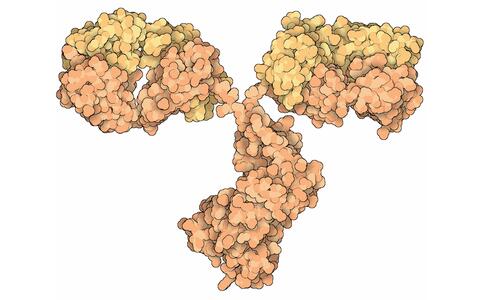
This is the actual shape of an antibody. The molecular model has a constant region “base” (in orange) and two variable region “arms” (in orange and yellow). The molecule is approximately 16 nanometers across.
“It is a miracle that blood cancer doesn’t develop more often”
These breaks are neither good or bad. “It’s again about finding the right balance,” explains the researcher. Nevertheless, she admits, it is astonishing how well the immune system’s control processes function: “It is a miracle that blood cancer doesn’t develop more often.”
There are certainly more than enough opportunities for this to occur. The body’s white blood cells multiply “like crazy” – they divide every few hours and have to copy their genetic material every time. According to Di Virgilio, this provides many opportunities for mistakes that could ultimately result in cancer. That’s not to mention the many DNA lesions that are introduced on purpose to reassemble antibody molecules.
Right at the end, once the researchers had already recorded their results, there was yet another surprise. In discussions with immunologist and B cell expert Professor Klaus Rajewsky from the MDC, they realized that ZMYND8 also “enhances” the antibody ability to recognize the antigen during immune responses. Di Virgilio points to the two arms of the antibody in her sketch. It appears that ZMYND8 also leads the aggressive enzyme to these variable regions of the antibody, where it causes random mutations to help improve its performance. “For the first time, we have described ZMYND8 as a factor that increases antibody diversity,” says Di Virgilio, calling this an “important process” that enables the organism to respond optimally to pathogens.
For Di Virgilio, this research is an example of the “winding road” of basic research. Ending up with different findings than expected, she explains, is part and parcel of the process. “That doesn’t mean we’re just taking stabs in the dark,” says the scientist. “We know a lot, but a lot is still a mystery.” Every research project is thus a “voyage into the unknown.”
Reference
Verónica Delgado-Benito, et al. (2018): „The Chromatin Reader ZMYND8 Regulates Igh Enhancers to Promote Immunoglobulin Switch Recombination.“ Molecular Cell. doi:10.1016/j.molcel.2018.08.042