Di Virgilio Lab
Genome Diversification & Integrity
Profile
Our Research Focus at a Glance
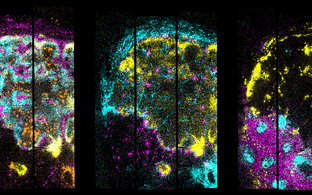
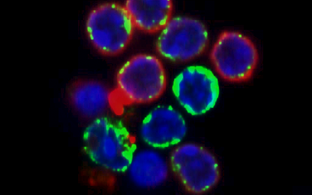
Accurate repair of both stochastic and programmed DNA double-strand breaks (DSBs) is a physiological and beneficial aspect of DNA metabolism since it ensures preservation of the genome on one side and the generation of diversity during meiotic recombination and lymphocyte receptor gene rearrangements on the other. Indeed, defective DSB repair is responsible for several human disorders characterized by developmental and neurological defects, cancer predisposition, and immunodeficiency.
Our group research aims at dissecting the molecular mechanisms that control the balance between antibody gene diversification and genome stability in mature B lymphocytes, and at translating our findings into general principles governing the ubiquitous preservation of genome integrity.