Immunotherapy for cancer: How tumor recurrence can be avoided
Since scientists discovered that there are immune cells that can in principle eliminate tumor cells, they have tried to use our immune system as a weapon against cancer. The underlying idea is simple although the implementation is sometimes difficult: in our body’s defense system, there are certain immune cells – T cells – that can detect and eliminate invaders like viruses or bacteria. Cancer cells can be identified as abnormal cells as well, but usually they are spared because T cells regard them as the body’s own cells.
T cells that can target tumors
With so-called T cell receptor (TCR) gene therapy researchers modify the T cells of a patient in vitro so that they can target cancer cells, and then put the T cells back into the patient. This has proven effective: in clinical studies, researchers were able to show that in patients with certain types of late-stage cancer, the tumors frequently regressed. However, this success was of short duration, since in many cases tumors recurred after treatment.
“Apparently the tumors are not recognized anymore by the T cells,” says Dr. Ana Textor. Textor is a postdoctoral researcher in the research group of Prof. Blankenstein at the MDC and the Charité, and is the lead author of the recent paper. “We wanted to find out how to shift the balance from cancer recurrence to cancer clearance,” says the biologist.
T cells patrol the body
The processes underlying immune response are complicated: T cells identify pathogens or malignant cells with the help of so-called antigens. These are foreign or abnormal proteins, e.g. proteins that are produced in a cancer cell as a result of a mutation. In the cell, these proteins are degraded by the proteasome into smaller fragments, which are then transported to the surface and presented on the outside of the cell.
It is only now that the foreign or abnormal cells can be detected. The T cells are constantly patrolling the body, scanning the surface of the cells for antigens. For this they have T cell receptors, a kind of antenna that can recognize the presented antigen fragments. The peptides of the antigens are called epitopes. Each T cell is highly specific: it is equipped with one T cell receptor type that can only recognize one specific epitope.
Searching for the best targets
Consequently, the mutated neo-epitopes presented on cancer cells are good targets for T cell therapy. “In particular, we wanted to know which parameters should be taken into account when choosing targets for TCR gene therapy to prevent tumor recurrence,” explains Textor.
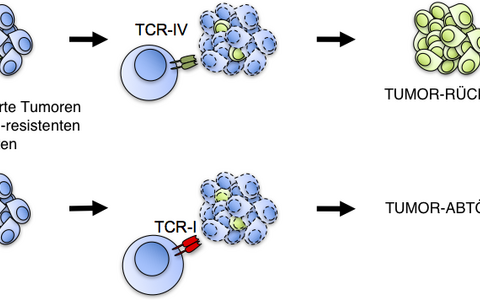
To find this out, she and her colleagues used large established tumors in mouse models for TCR gene therapy. They selected two very similar epitopes from a known model antigen that were both specific for the cancer cells. They were presented on the surface of the cells in comparable amounts and were easily recognized by the TCRs, which made them good candidates for TCR gene therapy.
The researchers produced in vitro T cells from the mice with the TCRs for each epitope and injected them into the bloodstream of the animals. The results were clear: while T cells against the one epitope eradicated the tumors, T cells against the other epitope induced tumor regression, which made the tumor subsequently recur.
A cell-signaling protein under suspicion
The researchers suspected another player in the immune system of being responsible for this: the interferon gamma. This is produced by T cells, and then stimulates the cancer cells and has in general an activating effect on immune response.
After being degraded by the proteasome, some epitopes have to be trimmed in the cell by an enzyme called ERAAP so that they can be transported to the surface of the cell. This enzyme is upregulated by interferon gamma. The researchers showed that the epitope that led to tumor rejection did not require trimming by ERAAP, while the epitope that led to tumor recurrence did.
And this is what explains the recurrence of the tumor in this model: in the large tumors most of the cells apparently responded to interferon gamma, presented both epitopes on the surface of the cells, and were attacked by the T cells. However, the cancer cells in the recurred tumors had been selected for unresponsiveness to interferon gamma.
Tumor cells unresponsive to interferon gamma can escape
When the large tumor was eliminated, these cells escaped from one of the two kinds of T cells. Without the interferon gamma, they were not able to present the epitope in sufficient numbers on the surface of the cells, and the T cells were therefore unable to detect them. These escaped cells grew into a new resistant tumor.
“Epitopes that do not require trimming by ERAAP are therefore probably a better choice for TCR gene therapy,” says Textor. Since the mouse model is well established, and it is known that many of the processes are similar in humans, the researchers assume that this result can be extrapolated to humans. Moreover, immune escape due to ineffective trimming by ERAAP has been described for HIV and CMV infections.
Accordingly, the new insights are an important step toward more successful application of TCR gene therapy, since they suggest that choosing targets that are independent of interferon gamma and ERAAP could prevent the recurrence of tumors.
The study was funded by the German Research Foundation (Sonderforschungsbereich-Transregio, SFB-TR36).
Ana Textor1, Karin Schmidt1,2, Peter‑M Kloetzel2,3, Bianca Weißbrich4, Cynthia Perez1, Jehad Charo1, Kathleen Anders1, John Sidney5, Alessandro Sette5, Ton N.M. Schumacher6, Christin Keller2, Dirk H. Busch4, Ulrike Seifert2,7, Thomas Blankenstein1,3,8 (2016): „Preventing tumor escape by targeting a post-proteasomal trimming independent epitope.“ Journal of Experimental Medicine. doi:10.1084/jem.20160636
1Max Delbrück Center for Molecular Medicine in the Helmholtz Association, Berlin, Germany; 2Institute for Biochemistry, Charité – Universitätsmedizin Berlin, Berlin, Germany; 3Berlin Institute of Health, Berlin, Germany; 4Institute for Medical Microbiology, Immunology and Hygiene, Technical University, München; 5La Jolla Institute for Allergy and Immunology, La Jolla, USA; 6The Division of Immunology, The Netherlands Cancer Institute, Amsterdam, Niederlande; 7Institute for Molecular and Clinical Immunology, Otto-von-Guericke- Universität, Magdeburg, Germany; 8Institute of Immunology, Charité – Universitätsmedizin Berlin, Germany.
Further information
- Press release on the same study.Oct 4, 2016
- Research Highlight on T‑cell gene therapy against cancer. April 6, 2016
Featured Image: T‑cells attack a cancer cell. Image: Alex Ritter, Jennifer Lippincott Schwartz and Gillian Griffiths, National Institutes of Health, CC-BY-NC.