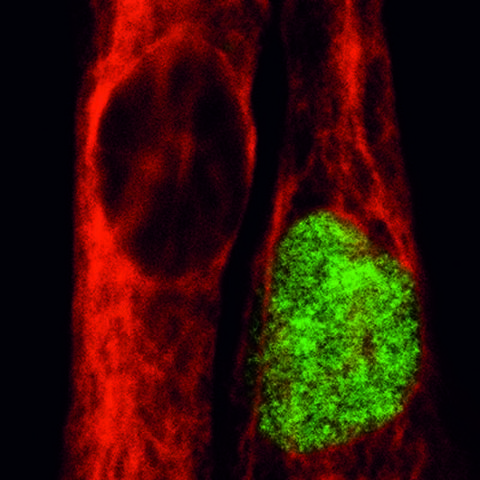
Tough ones – Human muscle stem cells
This is how Simone Spuler and Andreas Marg from the Experimental and Clinical Research Center (ECRC) have been treating human satellite cells for the first time. The process opens up new possibilities for the regeneration of defective skeletal muscles and the treatment of hitherto incurable muscle disorders.
You have subjected human muscle stem cells to a radical cure in the fridge. One would think that given such treatment cells would directly become comatose.
Simone Spuler: That is exactly what they do in principle. They enter into a kind of deep sleep mode. Colleagues from Paris observed in pathology that human muscle cells can be isolated from a corpse’s tissue even after death. This gave us the idea to preserve our human biopsy samples in the fridge.
What kind of cells are these and why is it so difficult to cultivate them?
Simone Spuler: Satellite cells are adult muscle stem cells, the number of which decreases with age. In contrast to pluripotent embryonic stem cells, which still can develop into different kinds of cellular tissue, these cells maintain only one program: to develop into a muscle fiber. Embedded between the cell membrane (sarcolemma) and the basement membrane, the muscle stem cell floats like a satellite outside the muscle fiber in an intermediate space, the stem cell niche. There it remains in an inactive state. It gets activated in the event of a muscle lesion, moves along the fiber to the site of the lesion, where it develops into healthy muscle fibers.
Andreas Marg: Researchers have been able to cultivate myocytes, the progenitor cells of skeletal muscle fibers, in vitro since 1971. However, they change their morphology and probably also their characteristics from one passage to the next. They pass over into a kind of geriatric state and lose their regeneration capability. So far it was not possible to use these cells in their original state for treatment; this is a problem affecting almost all stem cells, not only those in the muscles.
How did you solve the problem?
Simone Spuler: In the first instance we had a lot of patience and did not immediately trash the muscle fiber fragments, even if at first nothing could be seen in the cell culture. The actual trick is to leave the satellite cell in its natural environment, the stem cell niche. We chop up the human muscle biopsy samples, which we receive subsequent to the patients’ consent and that of the responsible office at the Ethical Review Committee, into muscle fiber fragments. That keeps the satellite cells’ “home”, the stem cell niche, intact. Preparing the fragments is analogous to a muscle injury in the body and gives the satellite cell the signal to start its regeneration program.
“I call this panic metabolism”
And then?
Simone Spuler: We put them into the fridge, complete with the fiber fragments. Coldness, no light, hardly any oxygen – a shock for the cell. Under such stress, the cell activates a survival program that remains as yet unidentified. I call this panic metabolism. We assume that the metabolism of the muscle stem cells reduces to a minimum.
Andreas Marg: Another positive effect of cooling is that all other cells in the muscle fiber fragments, such as connective tissue cells, do not survive this procedure. Only the muscle stem cells withstand it. After the period in the fridge we cultivate the fiber fragment at 37° C in the incubator. Within one to two weeks this develops into pure culture myocytes. Yet we are interested in what happens shortly before this development: the satellite cell activates its regeneration program and multiplies within the fiber fragment. That is exactly the moment when we transplant the muscle fiber fragments into a mouse. At the Charité CyberKnife Center, we irradiate a targeted muscle area of only a few millimeters in the animals. In doing so, that is, by destroying the mouse’s own satellite cells, we cause a local repair defect in the muscle. Moreover, the CyberKnife is a fantastic tool from the point of view of animal protection. We avoid full body irradiation, which would destroy the mouse’s immune system and make it sick.
How do you rate your results?
Andreas Marg: Promising. We not only see the development of new muscle fibers, but we can also prove that the human muscle stem cells massively multiply. The aim is to transplant the undifferentiated muscle stem cell and to use the full regeneration potential.
How do we know that the transplanted cells remain in their undifferentiated original state?
Andreas Marg: The various phases of satellite cell activation are identified by different marker proteins such as Pax7 and NCAM, which we verify. This allows us to say with certainty that a Pax7-positive cell has kept its stem cell potential and thus its regeneration capability. Morphologically I would not be able to easily differentiate between a Pax7-positive or negative cell.
You were successful with mice. What is the challenge with regards to humans?
Simone Spuler: Formal and institutional conditions are of essential importance. The human cells must be isolated under GMP (Good Manufacturing Practice) conditions before they may be used in humans. After all, this is a drug. A translational institute such as the ECRC could guarantee these conditions, provided it has the necessary logistic and financial support. Another challenge is to produce a sufficient amount of pure muscle stem cells to regenerate large muscles such as those in the upper thighs.
“Not so long ago and our only option was to give the patient a prescription for physiotherapy”
Is this not likely to take a long time, with only few people benefiting?
Simone Spuler: No, we are not talking about the distant future here. Already there would be the option of treating patients with a bad muscle regeneration rate following surgery or severe illnesses, for example, those who may have spent a long time in intensive care; this is a huge problem.
How have muscle disorders been treated so far?
Simone Spuler: Not so long ago and our only option was to give the patient a prescription for physiotherapy. I head the working group “Muscle Research” including the university outpatient clinic for muscle disorders. It is hard to watch patients becoming unable to walk, how they sit in a wheelchair and ultimately are not even able to breathe independently anymore and not being able to help. There are about 400 genetic muscle defects in addition to a number of acquired disorders. A lot has happened since the mid-1990s. We have learned to differentiate between these disorders and we already have researched many molecular causes. The first muscle disorders are now being treated with gene therapy in the context of international studies. Together with the working group headed by Zsuzsanna Izsvák we work on the genetic modification of satellite cells to make them available in the long term for an innovative form of treatment.
So what stem cell experts have failed to achieve so far, you have achieved with a fairly simple refrigeration method. Were you also sneered at?
Simone Spuler: Of course! ‘We don’t do voodoo here’ was one of the things we had to listen to. But our success redeems us. The main factor for this, among others, is the interdisciplinary collaboration of clinicians and basic research scientists in this project. Apart from Zsuzsanna Izsvák, we cooperate with the developmental biologist Carmen Birchmeier, who greatly supports us. A setting such as here in Berlin-Buch, with the Charité on the one hand and the MDC’s basic research on the other, and us, the ECRC, in the middle, you do not find often anywhere in the world.
Go to the original paper: Journal of Clinical Investigation
Follow this link for the joint press release by the Charité and the MDC.
Den ‘Author’s Take’ Video Abstract:
Featured Image: Well experienced team: Physician Simone Spuler and biophysicists Andreas Marg work together on muscle stem sells. Photo: Maimona Id, MDC