Tumors prevent immune cell maturation
Dendritic cells are very important for the body’s natural defense against viruses, bacteria and even tumor cells, because they uptake and process exogenous cell material, the foreign antigens, and present it to the immune system. However, only mature dendritic cells are able to perform this function and thus trigger a defensive immune response. If the cells do not mature, they foster immune tolerance instead. A team led by researchers Dr. Uta Höpken and Dr. Armin Rehm from the Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC) found that there is an astonishing number of immature dendritic cells in the vicinity of lymphomas and leukemias – cancers of the lymphatic system.
We were able to show in our experiments that mature dendritic cells have low C/EBPβ levels, while immature dendritic cells carry high levels of C/EBPβ.
“In an immature state, these cells actually support tumor growth by releasing certain signaling molecules,” says Höpken. Contact with tumor cells, he explains, results in the upregulation of the transcription factor C/EBPβ, which prevents the dendritic cells from maturing. This way, the tumor actively manipulates the immune system of the cancer patient to create more favorable conditions. This is demonstrated by the MDC team in their latest paper published in the U.S. journal Proceedings of National Academy of Sciences (PNAS), which is the result of a collaboration with other researchers including those from the University Hospital of Münster (UKM). The Experimental and Clinical Research Center (ECRC), a joint institution of the MDC and Charité – Universitätsmedizin Berlin, also participated in the study. This work was financed by the Wilhelm Sander Foundation.
Molecular switch keeps the balance
The microenvironment of lymphatic and blood system cancers has been a longstanding area of research at the MDC. To aid in this research, mouse models were created that can develop lymphomas and leukemias. Some time ago, Höpken and Rehm identified a particular protein, transcription factor C/EBPβ, as the crucial “molecular switch” that regulates the maturation of dendritic cells.
In their new study they show that the dendritic cells of transgenic mice that lack the gene for this protein mature in an uncontrolled way compared to the control animals. “From this, we concluded that the normal maturation and functionality of dendritic cells relies on this switch being regulated in its activity,” explains Höpken. A certain level of the switch protein is always present: “We were able to show in our experiments that mature dendritic cells have low C/EBPβ levels, while immature dendritic cells carry high levels of C/EBPβ.”
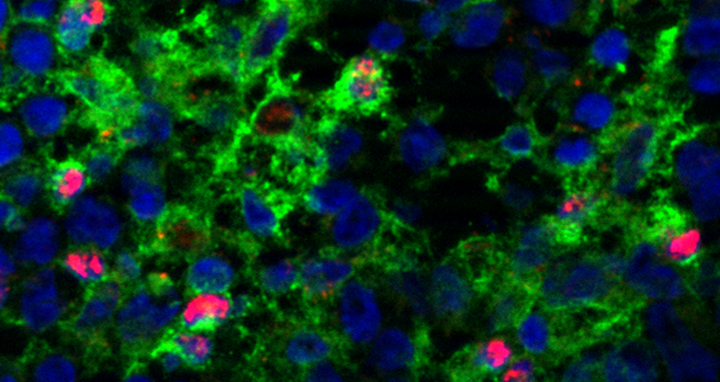
A team at the MDC has unraveled a previously unexpected role of the transcription factor C/EBPb in murine dendritic cell (in red) maturation and immunogenic functionality.
For a dendritic cell to be fully functional, it must go through time-regulated development from an immature to a mature stage: It is the immature cell that takes in the foreign antigens, and the mature cell that presents the processed antigen.
A complete lack of C/EBPβ led to the uncontrolled maturation of this cell type, leaving it only absorbing a reduced amount of foreign antigens. Tumor cells can actively intervene in this regulatory process. They block the body’s own immune defense by upregulating C/EBPβ and thus preventing maturation. “The time-regulated balance of this molecular switch’s activity is therefore crucial to the functional efficiency of these cells,” explains Höpken.
A reinterpretation of mTOR signaling pathway inhibition in cancer
It is not only dendritic cells that have this molecular switch. The same protein regulates the gene expression, maturation and proliferation of many other cells in the body – including those of the tumor itself. Drugs like the immunosuppressant rapamycin can virtually shut down C/EBPβ production. This is because an important part of the protein’s formation is regulated by the mTOR signaling pathway – which is blocked by the immunosuppressant.
“mTOR signaling pathway inhibitors are already used to treat cancers because of how effective they are at stunting cell growth. But until now, it was thought that this effect only impacted the cancerous cells or the vascular cells in the tumor,” says Rehm. “Based on our investigations, we assume that mTOR inhibitors also act on the immune cells in the immediate vicinity of the tumor via the same mechanism.” The suppression of C/EBPβ activity in dendritic cells therefore has two therapeutic effects. On the one hand, it prevents the release of signaling molecules and growth factors that promote lymphoma cells. On the other hand, maturation also supports the activation of other protective immune cells.
Tumor microenvironment as new target for cancer therapy
The microenvironment of the tumor – if our hypothesis is correct – unfortunately also fosters relapses.
“We know today that tumor cells cannot exist alone – not even very aggressive lymphomas. They need support from their immediate environment. And this need has long been underestimated,” emphasizes Rehm. “But the microenvironment of the tumor – if our hypothesis is correct – unfortunately also fosters relapses.” Even after successful chemotherapy, individual tumor stem cells and stromal cells that perform supporting functions in the tissue often remain. And it is here that the disease can flare up again. “It is possible that the growth of scattered tumor cells is supported by stromal cells and molecular switches such as the dendritic cell transcription factor C/EBPβ."
Stimulating maturation
Supporting the maturation of dendritic cells could be a complementary approach to cancer therapy. There are already drugs that improve the maturation status of these cells by docking to surface receptors, such as the known toll-like receptors. Accompanying immune-boosting treatment with such agents during chemo- or antibody therapy could therefore be very useful. “The problem of blocking the maturation of dendritic cells has been clinically recognized,” Rehm concludes. “We have therefore identified a new target and shown that it makes sense to intervene in this process. This provides impetus for drug and target screening by pharmaceutical companies.”
Text: Catarina Pietschmann
Further information
Lymphoma’s different route revealed
Literature
Florian Scholz et al.(2020): “The transcription factor C/EBPβ orchestrates dendritic cell maturation and functionality under homeostatic and malignant conditions,” PNAS, DOI: 10.1073/pnas.2008883117