Misfolding in the Alzheimer’s brain
Alzheimer’s develops secretly and silently. The disease begins its insidious attack on the brain years before the first symptoms appear. For reasons that are still unknown, small protein fragments known as beta-amyloid peptides undergo changes in their spatial structure. As a result, they become sticky and clump together between the neurons, or nerve cells, to form plaques. This triggers inflammation in the brain, where bundles of similarly misfolded proteins, the so-called tau fibrils, accumulate in the cells. Ultimately, the fibrils mean certain death for the cells. What exactly unleashes this deadly cascade, however, is still a big mystery to scientists.
Pathologically modified proteins affect healthy proteins
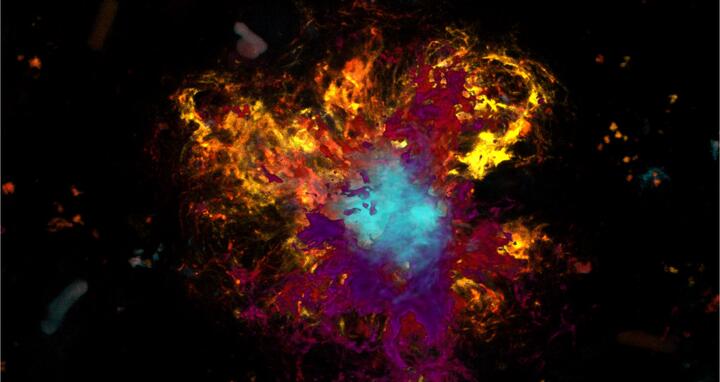
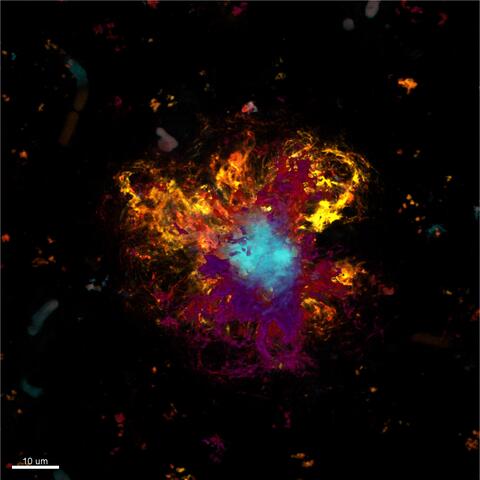
Beta-amyloid plaques are also found in the brain tissue of mice genetically modified to develop an Alzheimer’s‑like clinical picture. The core of the plaques is colored light blue, and the typical folded sheet-like structure is most pronounced here. Loose beta-amyloid enriched around the core region is shown in yellow and purple. Photo: Wanker Lab, Advanced Light Microscopy Platform.
“Only a few years ago did scientists discover that misfolded beta-amyloids are apparently capable of remodeling other proteins as well – similar to what can be observed in the prions, which were made widely known by mad cow disease,” says Professor Erich Wanker of the Berlin-based Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC). “If a misfolded peptide comes into contact with a healthy peptide, the spatial structure of the latter also begins to change,” explains the head of the Proteomics and Molecular Mechanisms of Neurodegenerative Diseases Lab. In this way, more and more of the harmful plaques are formed in the brain. Such properties have also been observed in the tau protein in experiments with mice. “Yet this does not mean that Alzheimer’s can be passed from person to person,” emphasizes Wanker. “The refolding occurs exclusively in the brain, from protein to protein.”
So far, it is unclear whether the misfolded amyloid and tau proteins can only transfer their disease-causing structure among their own kind or whether spreading – “seeding,” as Wanker calls it – can also occur between the two proteins. That is exactly what the MDC scientist and his team now want to investigate. Over the next three years, he will be supported in these efforts by the non-profit Alzheimer Forschung Initiative (AFI) as one of eleven researchers receiving AFI funding. The organization has awarded Wanker a grant worth €120,000. The largest private supporter of Alzheimer’s research at German universities and public institutes, the AFI has since 1995 been raising private funds to achieve it primary aim – to one day find a cure for Alzheimer’s.
The goal is to prevent the spread of amyloids in the brain
Prof. Dr. Erich Wanker
“I am delighted to receive this award, which will help me employ a research associate and also fund laboratory materials such as cell cultures and synthetically produced amyloid and tau proteins,” says Wanker. “Our goal in the next few years is to develop approaches for new drugs that stop misfolded proteins from proliferating and spreading in the brain.”
Initially, the researcher plans to do this by experimenting with neurons in the petri dish. “The cells will be genetically modified to reveal accumulations of misfolded proteins,” explains Wanker. “We will introduce these proteins into the cells and then examine whether the resulting aggregates are actually passed on from cell to cell.” Similar experiments are planned with fruit flies, i.e., in living organisms. “None of the Alzheimer’s drugs tested so far have targeted seeding,” says Wanker. And perhaps that is at least one of the reasons why there has been little success in treating the disease to date.
Text: Anke Brodmerkel
Further information
-
Project overview on the Alzheimer Forschung Initiative’s website (only in German)
-
Video presentations of the AFI-funded researchers (only in German)
Download
Beta-amyloid plaques are also found in the brain tissue of mice genetically modified to develop an Alzheimer’s‑like clinical picture. The core of the plaques is colored light blue, and the typical folded sheet-like structure is most pronounced here. Loose beta-amyloid enriched around the core region is shown in yellow and purple. Photo: Wanker Lab, Advanced Light Microscopy Platform. Image: AG Wanker, Technologieplattform „Advanced Light Microscopy“.
Contact
Jana Schlütter
Editor, Communications Department
Max-Delbrück-Centrum für Molekulare Medizin in der Helmholtz-Gemeinschaft (MDC)
Tel.: +49-(0)30 – 9406-2121
jana.schluetter@mdc-berlin.de oder presse@mdc-berlin.de
- The Max Delbrück Center for Molecular Medicine (MDC)
-
The Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC) is one of the world’s leading biomedical research institutions. Max Delbrück, a Berlin native, was a Nobel laureate and one of the founders of molecular biology. At the MDC’s locations in Berlin-Buch and Mitte, researchers from some 60 countries analyze the human system – investigating the biological foundations of life from its most elementary building blocks to systems-wide mechanisms. By understanding what regulates or disrupts the dynamic equilibrium in a cell, an organ, or the entire body, we can prevent diseases, diagnose them earlier, and stop their progression with tailored therapies. Patients should benefit as soon as possible from basic research discoveries. The MDC therefore supports spin-off creation and participates in collaborative networks. It works in close partnership with Charité – Universitätsmedizin Berlin in the jointly run Experimental and Clinical Research Center (ECRC), the Berlin Institute of Health (BIH) at Charité, and the German Center for Cardiovascular Research (DZHK). Founded in 1992, the MDC today employs 1,600 people and is funded 90 percent by the German federal government and 10 percent by the State of Berlin.