Stein Lab
Translational Oncology of Solid Tumors
Profile
Cancer metastasis is responsible for >90% of cancer deaths and remains a major treatment challenge. Our translational concepts aim at identification and exploitation of novel key molecules in metastasis for prognosis and therapy of solid cancers. Our group is devoted to thorough analysis of key drivers, their regulation, role in cancer cell signaling and functional impact in metastasis formation. In search of new drivers of metastasis we identified the novel, previously undescribed gene Metastasis-Associated in Colon Cancer 1 (MACC1) in human colorectal cancer (CRC) (Stein et al., Nature Med, 2009). MACC1 induces fundamental processes like proliferation, migration, invasiveness and metastasis in xenografted and transgenic mice. Meanwhile, MACC1 has been established by us and many other groups as key player, prognostic and predictive biomarker for tumor progression and metastasis in >20 solid cancers. Following our initial discovery of MACC1, >170 papers (PubMed) from groups worldwide were published until today, incl. meta-analyses on solid cancers, HCC and digestive system (Stein, Expert Opin Ther Targets, 2013; Mudduluru et al., Mech Mol Carcinogenesis, 2017). In recent years, we unveiled the transcriptional, post-transcriptional and post-translational regulation of MACC1. We discovered its transcriptional targets and protein-protein interactors, and unveiled them as new diagnostic, prognostic and predictive key players for tumor progression and metastasis. We generated the first transgenic MACC1 mouse models, discovering MACC1-induced cancer transition and its link to stemness. We identified the first transcriptional and post-translational small molecule inhibitors, restricting MACC1-induced metastasis formation in mice. Together with the metastasis inducer S100A4, which we first identified as Wnt-signaling target (Stein et al., Gastroenteroly, 2006; Dahlmann et al., Cancers, 2016), we generalized the impact of these biomarkers for early identification of high risk cancer patients. We exploited this knowledge for improved prognosis and response prediction using tumors and blood of solid cancer patients. Novel therapeutic approaches are currently tested in clinical trials to treat patients with metastatic disease using newly identified repositioned small molecule inhibitors acting on these biomarkers.
Team
Research
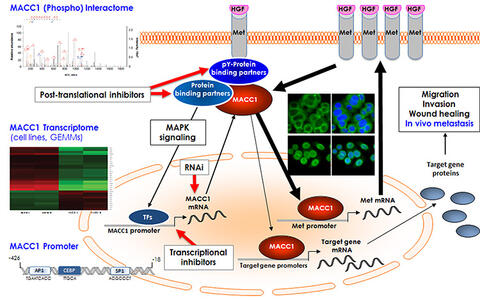
Key achievements for MACC1: MACC1 induces cell motility, proliferation, and scattering in vitro and metastasis in vivo. It acts as transcription factor regulating e.g. Met expression and interacts with proteins via e.g. its SH3 domain or proline-rich motif. By microarrays, we identified the MACC1 transcriptome. By HTS using the MACC1 promoter, we found first transcriptional MACC1 inhibitors. By MS, we identified the MACC1 interactome/phospho-interactome/ kinome for novel interventions. The MACC1 secretome was determined by SILAC.
- Cancer cell signaling of metastasis inducing genes, their networks and axes
In recent years, we unveiled key features of the MACC1 molecule, its expression regulation, and its post-transcriptional as well as post-translational regulation. To reveal MACC1 expression regulation, we identified and characterized the MACC1 promoter. The region from ‑426 to ‑18 constitutes the core promoter harboring functional motifs for binding of AP‑1, Sp1, and C/EBP transcription factors, which physically interact with the core promoter. Knock down of these transcription factors reduced MACC1 expression and MACC1 induced cell migration. In human colorectal tumors c‑Jun and Sp1 levels correlated to MACC1 expression and to metachronous metastasis (Juneja et al., Mol Oncol, 2013).
Another important aspect is the post-transcriptional regulation of MACC1. We explored the miRNA landscape for MACC1-induced metastasis. In silico analysis revealed MACC1 as potential target of miR-218, a tumor suppressor miRNA. We confirmed miR-218 as negative regulator of MACC1 expression, inhibiting MACC1 mediated migration, invasion and colony formation. Expression of these two molecules inversely correlated in a cohort of CRC tissues (n=59). Thus, miR-218 is post-transcriptionally inhibiting MACC1 expression and its metastasis-inducing abilities (Ilm et al. Oncotarget 2016).
To reveal, how MACC1 triggers expression alterations in cancer cells, we analyzed the MACC1‑induced transcriptome to track down key players mediating the MACC1-induced effects. We identified more than 1300 differentially expressed genes, including Spondin 2 (SPON2) with 90-fold overexpression. We demonstrated the binding of MACC1 to the SPON2 gene promoter. SPON2 overexpression in CRC cells increased metastasis in mice. CRC patients at stages I‑III, with high SPON2 expressing primary tumors, demonstrated 8 months shorter metastasis-free survival (MFS) compared with SPON2 low patients. Combining high SPON2 and MACC1 levels improved identification of high-risk patients with a 20 months shorter MFS, supporting MACC1 as trigger for other drivers of metastasis formation (Schmid et al., Oncogene, 2016).
As tumor cells are able to evade programmed cell death, we addressed the role of MACC1 in death receptor-mediated apoptosis. MACC1 knockdown sensitizes cancer cells to death receptor-mediated apoptosis and regulates Mcl‑1 and Fas expression via STAT signaling. MACC1 overexpression enhanced STAT1/3 phosphorylation and increased Mcl‑1 expression. Clinical relevance of Mcl‑1 regulation by MACC1 was shown by their positive expression correlation in patient tumors. Thus, we reveal a novel death receptor-mediated apoptosis regulatory mechanism in solid cancers through modulation of the STAT1/3‑Mcl‑1 axis by MACC1 (Radhakrishnan et al., Cancer Lett, 2017).
The Wnt/b‑catenin pathway is a key feature in cancer progression. We were first identifying the well-known metastasis inducer S100 calcium-binding protein A4 (S100A4) as a transcriptional target of Wnt/b‑catenin pathway. S100A4 is promoting EMT, migration, invasion, angiogenesis and metastasis. Its elevated expression in CRC and further cancer types correlates with poor prognosis. Post-transcriptional S100A4 regulation is not yet clearly elucidated. We showed that miR-505 – 5p and miR-520c-3p target the 3′-UTR of S100A4, inhibit its expression and S100A4 mediated migration and invasion. miR-520c-3p overexpressing CRC cells showed reduced liver metastasis. In a cohort of CRC patients (n=59), miR-520c-3p was downregulated. Our findings demonstrate that S100A4 is post-transcriptionally regulated by the tumor suppressers miR-505c-5p and miR-520c-3p, and miR-520c-3p expression is epigenetically silenced in CRC (Mudduluru et al., Oncotarget, 2017).
As chronic inflammation is known to promote tumor development and progression, we analyzed the S100A4-dependent link between inflammation, tumor progression and metastasis. We found that the acute-phase response proteins serum amyloid A (SAA) 1 and SAA3 are transcriptional targets of S100A4 via TLR4/NF-κB signaling. Expression of S100A4 and SAA1/3 in CRC patients correlates with reduced survival. SAA1/3 proteins are effectors of the metastasis-promoting functions of S100A4, and link inflammation to tumor progression (Hansen et al., Oncogene, 2015; coop Uni Copenhagen).
To decipher the S100A4 function in detail, we identified the receptor RAGE as mediator of S100A4-induced cell motility acting via MAPK/ERK and hypoxia signaling. RAGE by itself can serve as prognostic biomarker for CRC metastasis (Dahlmann et al., Oncotarget, 2014; DFG grant). In addition, we verified this finding of RAGE as mediator of pro-migratory response of extracellular S100A4 in human thyroid cancer (Medapati et al., Thyroid, 2015; coop Uni Manitoba).
- Preclinical models for understanding and intervening in tumor progression and metastasis
For translation of our achievements regarding MACC1 and S100A4 driven processes leading to tumor progression and metastasis formation, preclinical models are the essential link. We focused on generation of GEMMs and on use of cell line derived (CDX) and patient derived xenograft (PDX) mouse models. We generated the first GEMM for MACC1 with intestine-specific villin promoter (vil-MACC1) driven MACC1 overexpression. We crossed these mice with ApcMin mice. vil-MACC1/ApcMin mice (vs. ApcMin) showed increased number and size of tumors, and invasive phenotypes underlining the adenoma to carcinoma transition. This accelerated adenoma-carcinoma-sequence led to reduced survival of vil-MACC1/ApcMin mice (vs. ApcMin). Molecular analysis revealed increased Wnt and pluripotency signaling and a prominent upregulation of the pluripotency markers Oct4 and Nanog in the tumors of vil-MACC1/ApcMin mice. We demonstrated the binding of MACC1 to the Nanog gene promoter, which then regulates Oct4. We validated the MACC1 regulation of Nanog and Oct4 in CRC cells and found a strong correlation of MACC1 with Nanog and Oct4 in a CRC cohort of 60 tumors. This is the first evidence that MACC1-induced tumor progression in CRC acts, at least in part, via the newly discovered MACC1/Nanog/Oct4 axis (Lemos et al., Clin Cancer Res, 2016; Böck et al., Cancer Res, 2014; grants Wilhelm Sander foundation, Alexander von Humboldt foundation).
To evaluate the impact of MACC1, S100A4 or SPON2 on metastasis and to test intervention strategies, CDX with modulated expression of these genes were analyzed for metastasis formation (Stein et al., Gastroenteroly, 2006; Stein et al., Nature Med, 2009; Pichorner et al., 2012; Schmid et al., Oncogene, 2016, Mudduluru et al., Oncotarget, 2017). A variety of drug treatments targeting S100A4 with niclosamide, sulindac and calcimycin (Sack et al., J Natl Cancer Inst, 2011; Stein et al., Neoplasia, 2011; Sack et al., Mol Biol Cell, 2011) or targeting MACC1 with statins, rottlerin and MEK inhibitors (Juneja*/Kobelt* et al., Plos Biol, 2017; Kobelt et al., submitted 2018) showed effectiveness in reducing metastasis in mice.
As closer-to-patient models for testing drugs that efficiently intervene in metastasis formation, PDX of CRC were used. Particularly, PDX of CRC peritoneal metastasis were generated to evaluate biomarkers such as MACC1 and S100A4, to identify pathways for metastasis formation, and to predict response towards conventional and targeted therapies. In this context, we identified S100A4 as predictor for therapy response (E. Pachmayr, Berlin Cancer Society grant).
- Molecularly targeted intervention strategies for restriction and prevention of cancer metastasis
Although MACC1 is a key driver and biomarker for cancer progression and metastasis in a variety of solid tumor types, no MACC1 inhibitors were identified so far. We developed strategies for molecularly targeting MACC1- and S100A4-induced metastasis at transcriptional and post-translational level.
Transcriptional MACC1 inhibitors: To identify MACC1 transcriptional inhibitors we used human MACC1 promoter driven luciferase reporter CRC cells (Juneja et al., Mol Oncol, 2013) for high throughput screenings (HTS) employing the FMP-ChemBioNet library of more than 30,000 compounds (coop J.P. von Kries, FMP Berlin). The FDA-approved drug lovastatin and the small molecule rottlerin emerged as most potent MACC1 inhibitors. These drugs impaired binding of the transcription factors c‑Jun and Sp1 to the MACC1 promoter, thereby inhibited MACC1 transcription, and reduced MACC1 induced cell motility. In CRC-xenografted mice, lovastatin and rottlerin restricted MACC1 expression and liver metastasis. This is the first identification of inhibitors restricting metastasis via the novel target MACC1. This drug repositioning might be of therapeutic value for CRC patients (Juneja*/Kobelt* et al., Plos Biol, 2017; Mudduluru et al., Drug Resist Update, 2016).
Post-translational MACC1 inhibitors: As protein phosphorylation is a key feature for protein activity and functional regulation, we in silico predicted tyrosine phosphorylation (pY) sites in MACC1. pY mutated MACC1 was not able to induce motility and proliferation in vitro, and metastasis in vivo. Using mass spectrometry, we identified the interactome and the phospho-interactome of MACC1 (coop G. Dittmar, MDC; B. Küster, TU Munich), which is enriched for MAP kinase signaling components. Within the MACC1 kinome, we identified MEK1 as the only kinase able to phosphorylate MACC1 at tyrosines. MEK inhibitors reduced MACC1 induced cell motility. Combinatorial treatment with MEK1 inhibitors and statins synergistically reduced motility and proliferation in vitro. Remarkably, MACC1-induced metastasis in mice was restricted with FDA-approved MEK1 inhibitors such as AZD6244 (selumetinib) and GSK1120212 (trametinib). This shows that MAP kinase signaling is not linear leading to ERK activation, but branches at the level of MEK1. This fundamental finding of MACC1 being a newly identified MEK1 substrate opens new therapeutic interventions by targeting MEK1 for MACC1 stratified patients being at high risk for metastasis (Kobelt et al., submitted 2018).
Transcriptional S100A4 inhibitors: The S100A4 promoter-luciferase reporter was used for HTS using the NCI/NIH-Lopac library (coop R.H. Shoemaker, NIH/NCI, Frederick, MD). We identified the first S100A4 transcriptional inhibitors niclosamide, sulindac and calcimycin (Sack et al. J Natl Cancer Inst 2011; Stein et al. Neoplasia, 2011; Sack et al., Mol Biol Cell, 2011; Dahlmann et al., Methods Mol Biol, 2015; Dahlmann et al., Cancers, 2016). Niclosamide is currently combined with MACC1 inhibitors for more effective metastasis restriction in mouse models and is being applied in the clinical phase II trial NIKOLO (Burock et al., BMC Cancer, 2018; see 3.5).
- Clinical translation for improved prognosis, prediction and restriction of tumor progression and metastasis
Based on our discovery and validation of MACC1 as prognostic biomarker for CRC (Stein et al., Nature Med, 2009; Stein et al., Plos One, 2012; Nitsche et al., Ann Surg, 2012; Schmid et al., Mol Cancer, 2012), we refined the risk assessment by combined survival analyses of MACC1 expression with further molecular markers in a retrospective study of 99 CRC patients. Patients with high MACC1, mutated KRAS G13, wt BRAF V600 and MSS/MSI-low exhibited the highest risk for metachronous metastasis. This is the first report showing that identification of CRC patients at high risk for metastasis is improved by MACC1 in combination with KRAS G13 (Ilm et al., Mol Cancer, 2015).
Since tumor regions differ in function and expression, we addressed intratumoral heterogeneity by geographic expression of MACC1 in 187 CRC, correlated with clinicopathological and molecular features for improvement of survival prognosis. We found increased MACC1 expression from tumor center to front and overexpression in tumor buds as best predictors for metastasis formation and poor survival (Kölzer et al., BMC Cancer, 2015; coop A. Lugli, Uni Bern).
Mismatch repair (MMR) is an important parameter for therapy decision in CRC. We evaluated MACC1 expression to stratify stage II colon cancer patients with proficient MMR (pMMR) in 4 cohorts (Charité 1,2, BIOGRID 1,2) with 596 patients. Interestingly, pMMR/MACC1-low patients had a lower recurrence vs. pMMR/MACC1-high patients. 6.5% of all patients were pMMR/MACC1-low with no disease recurrence, resulting in a 17% higher 5‑year recurrence-free survival vs. pMMR/MACC1-high patients. Thus, MACC1 expression stratifies stage II colon cancer patients with unfavorable pMMR status. Very remarkably, pMMR/MACC1-low patients have a similar favorable prognosis to those with dMMR with implications of 5‑FU-based adjuvant therapy (Rohr et al., Ann Oncol., 2017; coop Roche Penzberg; Roche Basel; Ventana, Tucson AZ; Uni Melbourne; patent application: US62/422,573 15.11.2016).
In cross-entity approaches, we analyzed MACC1 for prognosis of patients suffering from glioblastoma multiforme (GBM). MACC1 increased the proliferative, migratory, and tumor-formation abilities of GBM cells. The c‑Met inhibitor crizotinib reduced MACC1-induced migration and tumor formation in organotypic hippocampal slice cultures of mice. In patient’ biopsies, MACC1 expression increased concomitantly with increasing WHO grade. MACC1 levels allowed discrimination of dormant and recurrent low-grade astrocytomas and of primary and secondary GBM. Strong MACC1 expression correlated with reduced patient survival (Hagemann et al., Neuro-Oncol, 2013; coop Uni Würzburg).
Curative treatment of hepatobiliary cancers is limited to surgical resection or liver transplantation. We proved MACC1 as decision-contributing biomarker in 156 patients with Klatskin tumors and intrahepatic cholangiocarcinomas. MACC1 expression of both entities was higher than in corresponding normal liver tissue. Klatskin tumor patients with a history of tumor recurrence had higher MACC1 than those without, linked to shorter overall and disease-free survival. The multivariate analysis confirmed MACC1 as independent factor for overall survival in Klatskin tumor patients (Lederer et al., Hepatology, 2015; BMWi grant).
Based on our established assay for circulating MACC1 and S100A4 transcripts in liquid biopsies of CRC patients, we exploited this assay for African American CRC patients (n=143). In this population CRC is highly aggressive with higher incidence. We assessed MACC1 protein in colon tissue and analyzed circulating MACC1 transcripts in patients with preneoplastic and neoplastic lesions in a prospective study. We conclude that MACC1 is expressed at early stages of colorectal oncogenesis and that the plasma transcripts can be used to stratify African American patients at risk for malignant colonic lesions (Ashktorab et al., J Transl Med, 2016; coop Howard Uni, Washington DC).
We determined circulating MACC1 and S100A4 transcripts in liquid biopsies for prognosis of gastric cancer patients (n=76) in a prospective study. Levels of circulating MACC1 transcripts were increased in these patients of each disease stage (vs. tumor-free volunteers). Patients with high circulating MACC1 transcript levels had shorter survival. Patients with high circulating MACC1 and S100A4 transcript levels had shorter survival vs. patients with both biomarkers low or with only one elevated biomarker (Burock et al., W J Gastroenterol., 2015).
To assess the plasma metabolites for stratification of stage II CRC patients according to metastasis risk, we performed metabolic profiling of plasma samples of non-metastasized and metachronously metastasized stage II CRC patients retrospectively (German population-based case-control multi-central DACHS study, n=92). Plasma metabolic profiles distinguish non-metastasized from metachronously metastasized stage II CRC patients, pointing to new diagnostic and prognostic tools (Zaimenko et al., Biochim Biophys Acta Rev Cancer, 2017; Zaimenko et al., submitted 2018; coop DKFZ/NCT Heidelberg).
Apart from determination of the diagnostic, prognostic and predictive value of MACC1 and S100A4, clinical translation of our preclinical findings on metastasis inhibition by repositioning of small molecule drugs is currently underway. We use the FDA-approved anti-helminthic drug niclosamide in the clinical phase II trial NIKOLO: Treatment of stage IV CRC patients using niclosamide for restriction of tumor progression and metastasis (EudraCT 2014−005151−20, NCT02519582; coop U. Keilholz, S. Burock, Charité). Drug-induced S100A4 modulation is monitored for therapy response using our blood-based S100A4 assay together with HPLC for niclosamide in patient blood (Burock et al., BMC Cancer, 2018). Our patent on niclosamide repositioning as anti-metastatic drug (EP2699319) was granted (9.8.2017).