Unravelling the drivers of type 2 diabetes
About 537 million people globally had type 2 diabetes in 2021 and the disease burden is only projected to grow. Type 2 diabetes occurs when beta cells, which reside in so-called islets in the pancreas, cannot produce sufficient insulin to ensure the uptake of sugar from the blood into tissue cells after meals. Although there are drugs that stimulate the release of insulin, they frequently fail long-term, and patients eventually must inject insulin.
“We know that our current ways of pharmacologically intervening by stimulating insulin secretion don’t lead to long-term reversal of diabetes. It’s like putting your foot on the gas pedal and then keeping it on, and at some point, the beta cells fail,” says professor Maike Sander, scientific director of the Max Delbrück Center. Together with professors Kyle Gaulton, University of California San Diego, and Sebastian Preissl, University of Freiburg, she is a senior author of the study published in “Nature Genetics.”
Scientists are studying the drivers of diabetes to make better drugs. The U.S. National Institutes of Health funded Sander and her collaborators to understand how gene regulation can predispose people to type 2 diabetes. Now, the team has found that a switch in the relative abundance of beta cell subtypes is associated with type 2 diabetes.
A more detailed picture of pancreatic islets
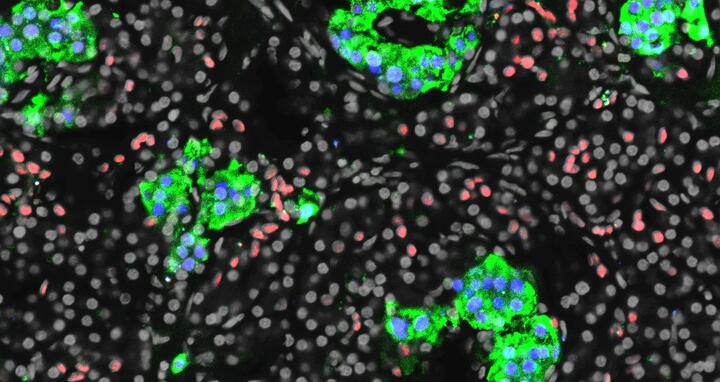
Section of a human pancreas stained for insulin in green, the beta cell marker PDX1 in blue, and the ductal cell marker CDX2 in red.
Due to technological limitations, scientists used to study pancreatic islets in bulk to understand disease progression. But this did not give them a detailed picture as islets contain various types of endocrine cells, and even the beta cells have two subtypes with unique functional and gene regulatory profiles. One beta subtype secretes more insulin when exposed to glucose than the other.
Sander and her colleagues harnessed the power of novel single-cell technologies to simultaneously analyze gene regulation and function of individual beta cells and learn how these features change on the path to type 2 diabetes.
The scientists analyzed islet samples from 34 deceased organ donors who were either not diabetic, had pre-diabetes or type 2 diabetes. They mapped the gene regulatory changes and then correlated these with changes in beta cell function, which they evaluated by analyzing individual beta cells for their ability to secrete insulin in response to glucose.
This switch is a hallmark of type 2 diabetes.
They found that in type 2 diabetes, the relative proportion of the two beta cell subtypes was flipped; the subtype that is less common in non-diabetic individuals was more prevalent in people with the disease. “This switch is a hallmark of type 2 diabetes,” Sander says.
“The subtypes did not switch in people with pre-diabetes,” Sander says. “It suggests one already has to have elevated blood glucose levels for the subtype switch to be present.”
They also found an indication that in people who are genetically predisposed to developing type 2 diabetes, there is a higher propensity for the beta cell subtype switch to occur.
Drug screening with organoids
“This feature of diabetes should be studied further as it could potentially be leveraged to develop new drugs,” Sander says. “If one could prevent the subtype switch, could one prevent progression to type 2 diabetes? That is the key question.”
At the Max Delbrück Center in Berlin, her lab is now developing organoids that model pancreatic islets as a screening tool to find drug targets. “This will allow us to conduct screens for genes and drugs and test effects on the ability of beta cells to secrete insulin,” Sander says.
gav
Further information
- Gaulton Lab at UCSD
- Preissl Lab, University of Freiburg
- Organoid research at the Max Delbrück Center
Literature
Gaowei Wang et al (2023): “Integrating genetics with single-cell multiomic measurements across disease states identifies mechanisms of beta cell dysfunction in type 2 diabetes.” Nature Genetics, DOI: 10.1038/s41588-023 – 01397‑9