Finding the source of the leak
Clinicians, geneticists and molecular biologists, including from the Max Delbrück Center for Molecular Medicine (MDC) and the Charité University Hospital in Berlin, came together to track down the specific gene involved in albuminuria – a condition where a protein called albumin leaks out of blood and into urine via the kidneys.
Albuminuria is a symptom of kidney damage. It usually indicates that a specific filtration barrier within the kidneys is not functioning properly. Instead of only allowing small liquid waste molecules to pass through, larger molecules that should stay in the blood also escape.
You need input from all different type of groups and researchers to actually push things farther.
To determine possible genetic contributors to this condition, the researchers used a variety of methods drawing on basic research using rats and zebrafish, advanced imaging and genetic tools, as well as clinical work with human patients. “The combination of different approaches is what I find cool and fascinating about this work,” Dr. Daniela Panáková, who heads MDC’s Electrochemical Signaling in Development and Disease lab. “You need input from all different type of groups and researchers to actually push things farther.”
Narrowing down
A key region on a particular chromosome in rats had previously been identified as associated with albuminuria. Researchers were able to narrow this region further through a process called congenic substitution mapping, where they compare the effects of precise DNA changes in two rat models, one susceptible and one resistant to albuminuria. But there were still an estimated 75 possible candidate genes.
Messenger RNA, which codes for DNA, helped unlock the mystery. By sequencing messenger RNA taken from glomeruli, the blood vessel structures within the kidneys responsible for filtering blood, the researchers were able to narrow the list down to ten genes. Analyzing which gene was expressed more during onset of albuminuria revealed the answer: gene tmem63c, which codes for transmembrane protein TMEM63C.
Not just rats, but zebrafish and humans too
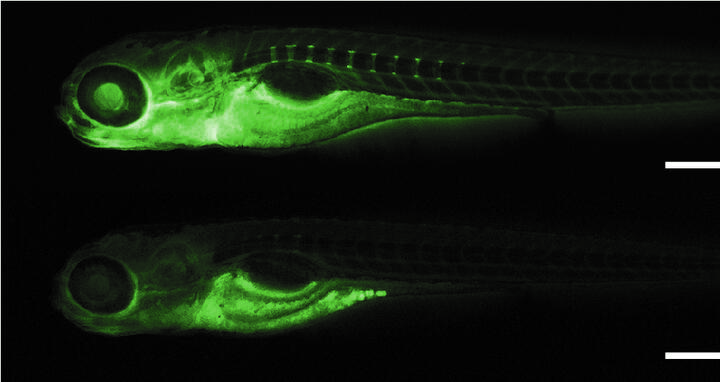
To confirm that this gene was indeed contributing to albuminuria, Panakova’s team tested the function of the gene in zebrafish. When they turned off the gene, the zebrafish would leak a florescent green molecule similar in size to albumin out of their kidneys. Instead of the molecule staying in the blood stream, causing the entire zebrafish to appear florescent green, it was discharged. This provides strong evidence that the gene, and its related protein, does impact the glomerular filtration barrier, a highly specialized blood filtering system within the kidneys.
MDC researchers were able to demonstrate the functionality of the tmem63c gene using zebrafish. Zebrafish with the gene have a green fluorescent molecule throughout their blood stream (top), while a zebrafish with the gene turned off (bottom) has lost fluorescence, indicating the molecule has leaked out of the blood stream via the kidneys.
The group further investigated to see what kind of damage was occurring when this gene was turned off. The filtration barrier has three main layers; they found that the cells in the top layer, called podocytes, were misaligned where they attach to the middle layer, called the basement membrane. This ultimately led to the fluorescent protein leaking out.
The filtration barrier functions similarly across species
When they added mRNA for tmem63c protein from rats, the leakage stopped. This indicates that not only is this gene directly involved with the proper functioning of the filtration barrier, but that it functions similarly in rats, zebrafish and humans.
Indeed, patients with scarred glomeruli, a disease that is known as focal segmental glomerulosclerosis, had a significant decrease of tmem63c expression, compared to healthy control subjects, and most had almost no TMEM63C proteins in their glomeruli. Further analysis of human podocyte cells showed impaired viability when the gene was turned off.
“Genetic conservation between zebrafish and other vertebrates is more or less well known,” said Nicola Müller, a Ph.D. student at MDC who co-authored the paper. “But our studies nicely prove the genetic conservation of tmem63c across species and identifies the zebrafish as a valid animal model for further research on a possible molecular mechanism of TMEM63C.”
Next steps to understand the role of the protein
While the protein has been identified and verified, there is still much more work to be done.
“We know that it is important, but we still don’t understand on a cellular level why it is important,” Panáková said. “We want to understand on a cellular and molecular level what is wrong when the protein is not functioning properly.”
The group plans to continue drilling down further to understand its role. Others in the same family of transmembrane proteins can act as channels, links or scaffolds. This sort of insight could help inform development of potential therapies to treat patients with damaged kidneys, as well as potentially address loss of function during normal aging processes.
Text: Laura Petersen
Original Publication
Angela Schulz, Nicola Victoria Müller et al. (2019): „Analysis of the genomic architecture of a complex trait locus in hypertensive rat models links Tmem63c to kidney damage“, eLife. DOI: 10.7554/eLife.42068