📺 A molecular motor for membrane constriction
Every cell is protected against external influences by a membrane. But in order to engulf nutrients or other substances, a part of the cell membrane folds in, enclosing the tiny particles and fluids that the cell needs in a vesicle. The vesicle eventually breaks off from the membrane and enters the cell. This process is known as endocytosis. Some viruses also gain access to the cell interior via this route.
It has been known for some time that the protein dynamin acts as a molecular motor that drives vesicle constriction. Professor Oliver Daumke, head of the Structural Biology of Membrane-Associated Processes Lab at the Berlin-based Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC), began studying the inner workings of this cellular machine about ten years ago. Now, in collaboration with researchers from the Fritz Haber Institute of the Max Planck Society (FHI) in Berlin and the Weizmann Institute of Science in Rehovot, Israel, the structural biologist has succeeded in creating a computer model that provides an exhaustive description of how the motors function in the cell membranes.
Crowning achievement of years of dynamin research
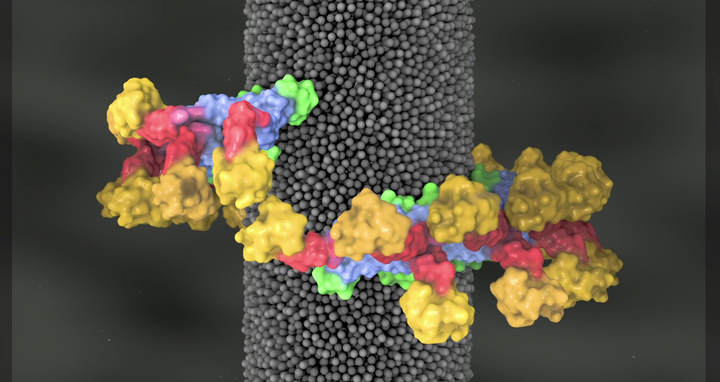
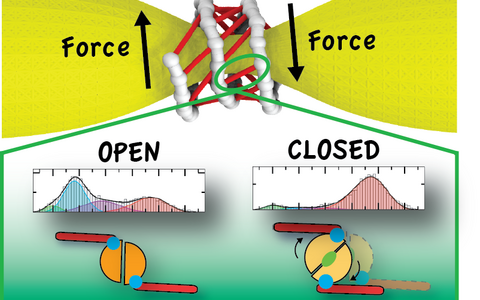
Dynamin coils around the neck of a vesicle (yellow) to form a right-handed helix (gray). In the GTP-bound structure, proteins from one rung of the helix form contacts with proteins in the adjacent rung (red dashes). Driven by GTP hydrolysis, the dynamin motor switches from an open to a closed state – these states can be observed spectroscopically (bottom). The transition between these states leads to the creation of a force that pulls the helix together like a snare.
“The study is the crowning achievement of our many years of research into dynamin,” says Daumke. The scientists had already shed much light on the protein’s structure and several of its states. “But it wasn’t until now that we gained a real understanding of how this cellular machine manages to pinch off membrane vesicles from the rest of the cell membrane,” reports Daumke. The study is been published in the journal “Proceedings of the National Academy of Sciences of the United States of America”, or PNAS for short.
The lead author of the study is Dr. Oleg Ganichkin, a biochemist and molecular biologist who conducted spectroscopic experiments at the MDC and the Weizmann Institute to detect the different states of dynamin. The last author is Dr. Jeffrey Noël, a physicist who did research in Daumke’s lab as well as at FHI. “We were very fortunate to be able to recruit such a great team for this study,” says Daumke. It was, he adds, a big plus that Noël was able to feed Ganichkin’s experimental data directly into the computer simulation. “Our model therefore works with realistic forces as they actually occur in the cell membranes,” Daumke says. “It can now be used to explain much of the data that we and other labs obtained in previous experiments.”
A helix that constricts during endocytosis
As the team led by Daumke and Noël reports, about 40 to 50 dynamin molecules work together in a helix-shaped structure. “Its shape resembles the spring in a ballpoint pen,” explains Daumke. The dynamin molecules arrange themselves in pairs within the spring-like helix. These dimers move in a kind of paddling motion, using energy obtained from the hydrolysis of the fuel molecule GTP. “In the process, the helix coils up tighter and tighter, causing the cell membrane to become increasingly thin at the point enclosed by the spring structure,” explains Daumke. This continues, he says, until the vesicle breaks off and enters the interior of the cell.
The new knowledge about the detailed workings of the dynamin motor may one day be useful in medicine. “We know, for example, that centronuclear myopathy, a form of muscle weakness, results from a mutation in the dynamin gene,” says Daumke. The gene mutation causes the protein to become overactive and act as a molecular motor not only on the outer cell membrane, but also on membranes in the cell interior that surround the myofibrils in a muscle cell like a well-fitted cuff. “The overactive dynamin damages this cuff, which is why the muscle cells no longer work properly,” explains the structural biologist.
Simulations were used to model the constriction of the dynamin helix. The example shows a simulation for a very long dynamin helix. The authors were, however, able to show that shorter dynamin helices, such as those found at the neck of membrane vesicles in the cell, also generate enough force to constrict the membrane neck.
How do mutations in dynamin affect the motor?
Daumke believes he has reached the end of the road on dynamin research. “We have answered the big questions, and all the knowledge we have acquired over the past ten years is summarized in the current study,” he says. Yet the MDC scientist is not entirely sure: “Perhaps we will soon explore how to repair mutant dynamin so that it works properly.” For people suffering from centronuclear myopathy, such research would offer a glimmer of hope.
Text: Anke Brodmerkel
Further information
Press release: “Keeping a cell’s powerhouse in shape”
Literature
Oleg M. Ganichkin et al. (2021): „Quantification and demonstration of the collective constriction-by-ratchet mechanism in the dynamin molecular motor”. PNAS, DOI: 10.1101/2020.09.10.289546