Fat tissue fuels gut inflammation
Crohn’s disease is a chronic inflammatory disorder of the intestinal tract affecting millions of people worldwide. One prominent feature of the disease is “creeping fat” — adipose tissue that wraps around inflamed sections of the intestine. Scientists have long suspected that this fat tissue may influence disease progression, but the underlying mechanisms have remained unclear
Now, researchers from Charité – Universitätsmedizin Berlin, the Berlin Institute for Medical Systems Biology at the Max Delbrück Center (MDC-BIMSB) and their collaborators show that fat tissue releases signals that ultimately damage the intestinal barrier in Crohn’s disease. They also show that acquired genetic mutations in immune cells may fuel the disease.
“There is a longstanding link between obesity and Crohn’s disease,” says Dr. Ashley Sanders, Group Leader of the Genome Instability and Somatic Mosaicism Lab at the Max Delbrück Center and a senior author of the paper. “While it’s fairly well established that adipose tissue secretes molecular signals that promote inflammation, here we detail the link between fat-derived signals and intestinal inflammation.”
Mice with no fat tissue
The researchers first conducted experiments in a mouse model of generalized lipodystrophy – an extremely rare disease in which affected people lack fat tissue among other symptoms.
When they exposed the mice to a chemical substance known to cause intestinal inflammation, they found the lipodystrophy mice were more resistant than normal mice. Their intestinal barrier remained stable and they had low levels of pro-inflammatory immune cells, particularly Th1 and Th17 T cells – specialized subsets ofimmune cells that coordinate immune responses.
When the team transplanted adipose tissue back into the lipodystrophy mice, they became more susceptible to the chemically induced gut inflammation. Remarkably, they observed this effect only when the transplanted fat was capable of producing leptin, suggesting that leptin plays a central role in regulating the immune system in mice.
“These findings were expected because previous research had already established that fat tissue is an active immune organ,” says Dr. Britta Siegmund, a senior author of the paper at Charité — Universitätsmedizin Berlin. “But they nevertheless highlight that signals derived from adipose tissue can substantially amplify intestinal inflammatory responses.”
Puzzling clinical findings
However, the results appeared to conflict with findings from a clinical case at Charité: A patient with acquired generalized lipodystrophy who had developed Crohn’s disease, despite having no fat tissue at all. “This was curious and we wanted to understand why this patient had developed the disease,” Siegmund adds.
Using single-cell sequencing, the Sanders lab examined immune cells extracted from the patient’s blood and intestinal tissue. The analyses showed an unusually large group of cells that carried a mutation in a gene called NRAS, which helps regulate cell growth and survival. This suggested that these cells had multiplied from a single original cell in a process called clonal expansion – such mutated cells arise spontaneously and are not inherited. Cells with mutated NRAS survive longer and divide more than they should, potentially driving chronic inflammation.
“This mutation likely helped the inflammatory T cells survive and expand over time,” explains Sanders. “That persistent expansion may have fueled the systemic and intestinal inflammation that was observed. What’s more, our results show how powerful single-cell genomic technologies can be for uncovering hidden genetic changes in individual immune cells,” she adds.
Toward more personalized treatment
Among other pro-inflammatory signals, adipose tissue also secretes mediators called adipokines — including leptin, which is best known for regulating appetite and metabolism. In the study, the lipodystrophy patient was given a synthetic form of leptin to treat type-2 diabetes. While the hormone improved the patient’s metabolic health, it also increased pro-inflammatory signaling and raised levels of Th17 immune cells.
“The finding highlights how adipokines such as leptin and genetic changes in immune cells interact to influence chronic inflammatory diseases,” says Dr. Carl Weidinger, another senior author of the study at Charité. This dual role of metabolic and genetic factors could help explain why Crohn’s disease behaves differently in different people, he adds. “Understanding these interactions may help us develop more precise, personalized approaches to treatment.”
Text: Gunjan Sinha
Further information
Literature
Marilena Letizia, Toka Omar, Patrick Weidner et al. (2026): “Characterization of intestinal immune responses in generalized human and murine lipodystrophy.” Journal of Clinical Investigation, DOI: 10.1172/JCI192322.
Image
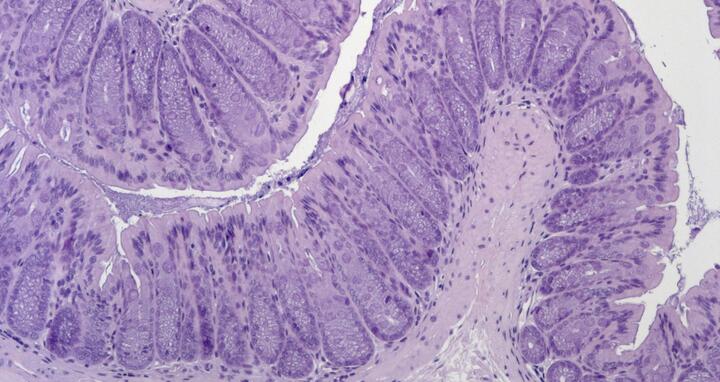
Colon tissue from a mouse model of acquired generalized lipodystrophy. Colon tissue remains intact after exposure to a chemical that causes gut inflammation. © Anja Kühl, Charité
Contacts
Dr. Ashley Sanders
Group Leader
Genome instability and somatic mosaicism
Max Delbrück Center
+49 30 94061426
Ashley.Sanders@mdc-berlin.de
Jana Schlütter
Editor, Communications and Marketing
Max Delbrück Center
+49 30 9406 – 2121
jana.schluetter@mdc-berlin.de or presse@mdc-berlin.de
- Max Delbrück Center
The Max Delbrück Center for Molecular Medicine in the Helmholtz Association lays the foundation for the medicine of tomorrow through our discoveries of today. At locations in Berlin-Buch, Berlin-Mitte, Heidelberg, and Mannheim, interdisciplinary teams investigate the complexity of disease at the systems level – from molecules and cells to organs and entire organisms. Together with academic, clinical, and industry partners, and as part of global networks, we turn biological insights into innovations for early detection, personalized therapies, and disease prevention. Founded in 1992, the Max Delbrück Center is home to a vibrant, international research community of around 1,800 people from over 70 countries. We are 90 percent funded by the German federal government and 10 percent by the state of Berlin.