Gene therapy: T cells target mutations to fight tumors
The efficacy of the immune system to combat disease makes the body’s own healing powers a promising approach in the fight against cancer. A Berlin research group led by Wolfgang Uckert, Thomas Blankenstein and Matthias Leisegang has been collaborating with U.S. researcher Hans Schreiber in a project to use immune components called T cells to target mutant proteins produced by cancer cells.
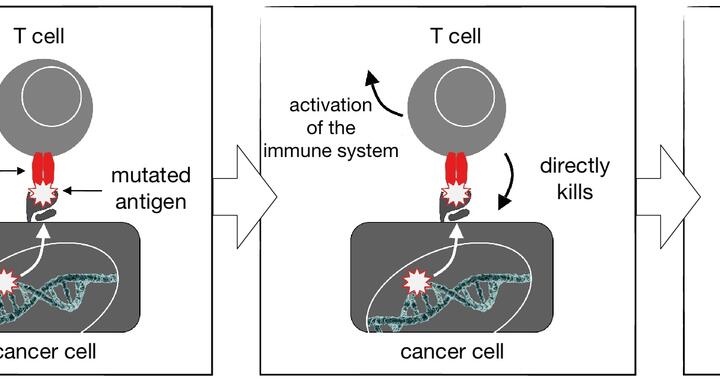
T cells are “special forces” of the immune system that patrol the body and are equipped with T cell receptors molecules. The receptors scan the surfaces of all body cells for protein fragments called antigens. Antigens are produced within cells and are usually derived from pathogens. If their foreign origins are recognized, T cells will kill these cells. Antigens may also arise from abnormal proteins resulting from mutations in the genes of cancer cells, which may also be recognized as “foreign” by T cells.
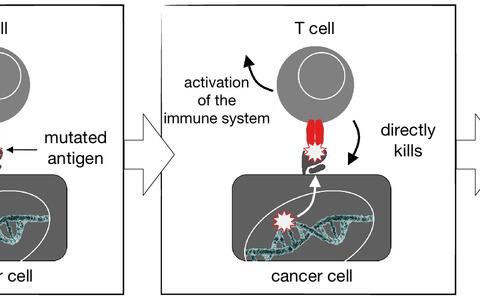
Even though T cells migrate into tumors and potentially can recognize their antigens, the defense mechanism seems to fail during the formation of tumors. T cells in the tumors are usually inactive and therapeutically almost useless, Matthias Leisegang says. “But it is possible to obtain fresh T cells from a patient’s blood and transfer tumor-specific T cell receptors into them,” Leisegang says. “The transfer of the T cell receptor is carried out using genetically modified and functionally inactivated viruses that can insert their genetic material into millions of T cells. When these modified cells are infused into the patient, they are able to fight the tumor.”
Hans Schreiber’s research team at the University of Chicago analyzed gene therapy with T cell receptors in mice. The scenario mirrored the situation that would be faced in the clinic. First the scientists analyzed the genes of an individual mouse tumor. They identified a mutation that occurred in all regions of the tumor and also found its way to the surface as an antigen. T cells were taken from the mouse, armed with a T cell receptor that would target this antigen, and were then administered to the animal. The cells almost completely destroyed the tumor. However, T cell therapy had to be combined with local radiation to eliminate the tumor in the long term. The results of the study were published in the journal Clinical Cancer Research.
Now, in a publication in the Journal of Clinical Investigation, the Berlin researchers show the importance of preliminary animal studies to ensure the final success of mutation-specific therapies. Even if some mutations appear to be suitable targets for therapy after tests in cell cultures, they can have disappointing results in the real context of a patient’s body. The research group of Thomas Blankenstein and Wolfgang Uckert were able to analyze the antigens and clearly distinguish between “good” and “bad” T cell targets by using a humanized mouse model. “This means that we have developed an animal model to test the therapeutic suitability of T cell receptors and antigens, which is an important prerequisite for clinical applications,” Leisegang said.
Although the efforts of Matthias Leisegang and his colleagues to train immune cells to fight cancer by targeting mutations were successful in mice, he emphasized that the patient individualized treatment is not yet ready for use in humans. Clinical tests based on similar methods are currently underway, but so far targeting only antigens without mutations. In those cases, the T cells may also attack healthy tissue. Targeting cancer-specific mutations, the researchers say, would cause fewer side effects. But the new method is also much more complex because it has to be individualized for each individual patient. With support from the BIH, the groups are now working with other teams at the MDC and Charité to apply their research to the clinic.
The project is funded by the BIH (CRG‑1) and the German Research Foundation (DFG, SFB TR36). A Visiting Fellowship from the Einstein Foundation made Hans Schreiber’s research stay in Berlin possible.
Matthias Leisegang1, Boris Engels2, Karin Schreiber2, Poh Yin Yew3, Kazuma Kiyotani3, Christian Idel2, Ainhoa Arina2, Jaikumar Duraiswamy2, Ralph R. Weichselbaum4, Wolfgang Uckert5,6, Yusuke Nakamura3 and Hans Schreiber1,2 (2015): “Eradication of large solid tumors by gene therapy with a T cell receptor targeting a single cancer-specific point mutation.” Clinical Cancer Research 22(4). doi:10.1158/1078 – 0432.CCR-15 – 2361
1Institute of Immunology, Charité, Berlin; 2Department of Pathology, University of Chicago, USA; 3Department of Medicine, University of Chicago, USA; 4Department of Radiation and Cellular Oncology, Ludwig Center for Metastasis Research, University of Chicago, USA; 5Molecular Cell Biology and Gene Therapy, Max Delbrück Center for Molecular Medicine, Berlin; 6Institute of Biology, Humboldt-Universität Berlin.Matthias Leisegang and Boris Engels contributed equally to the work.
Matthias Leisegang1,2, Thomas Kammertoens2, Wolfgang Uckert1,3 and Thomas Blankenstein2,4 (2016): “Targeting human melanoma neoantigens by T cell receptor gene therapy.” Journal of Clinical Investigation. doi:10.1172/JCI83465.
1Molecular Cell Biology and Gene Therapy, Max Delbrück Center for Molecular Medicine, Berlin; 2Institute of Immunology, Charité, Berlin; 3Institute of Biology, Humboldt-Universität Berlin, 4Molecular Immunology and Gene Therapy, Max Delbrück Center for Molecular Medicine, Berlin. Wolfgang Uckert and Thomas Blankenstein contributed equally to the work.
Contact
Josef Zens
Press officer
+49 30 9406 – 2118
presse@mdc-berlin.de