Don’t underestimate the power of the dark side: Understanding the dark zone in B cell differentiation
Scientists at MDC have revealed the importance of a transcription factor pathway in B cell maturation. The activation of mature B cells is required for normal immune system function but errors in the process can cause cancer. “The transcription factor FOXO1 was the main focus of our analyses,” reports Dr Sandrine Sander, “and we found that it is a key element in B cell-mediated immune responses.”
B cells are white blood cells that produce antibodies to fight infections. When the immune system recognises a foreign target, B cells are activated. The maturing B cells form a germinal centre in lymphoid tissues such as the spleen or tonsils. Here they undergo a germinal centre reaction, where the B cells modify their genome so that they can generate antibodies that efficiently combat a specific infection.
Dr Sander and her colleagues from Prof Klaus Rajewsky’s group at MDC are interested in how B cells respond to pathogens, including the mechanisms of germinal centre development. In a recent study the scientists focused on the transcription factor FOXO1 and how it contributes to B cell differentiation in the germinal centre. They investigated FOXO1 because it is frequently mutated in patients suffering from certain cancers – lymphomas originating from germinal-centre B cells.
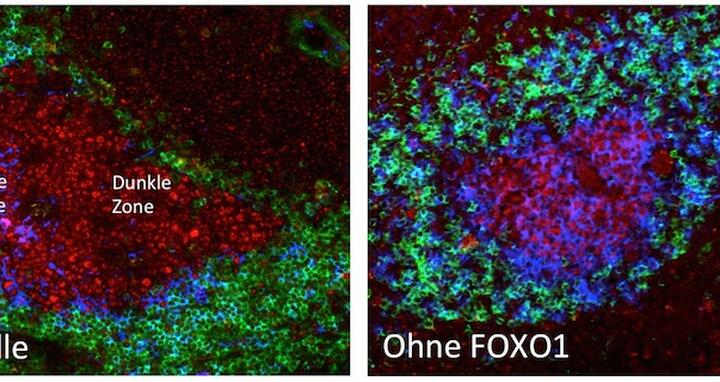
To investigate the role of FOXO1 in B cell maturation, the researchers studied mice where the FOXO1 gene was specifically deleted in germinal-centre B cells. This allowed them to work out how FOXO1 affects the development of these cells. They inspected the lymphoid tissues of the mice and found that germinal centres formed, but had an abnormal structure. The germinal centres lacked the usual ‘dark’ zone, which gets its name because it is so densely packed with dividing B cells that it looks dark under a microscope.
“We demonstrated that FOXO1 is a key element for the formation of the dark zone which means it is decisively involved in immune defence,” Dr Sander and Prof Rajewsky emphasise.
Germinal centres normally have two zones with separate functions. The dark zone is where maturing B cells undergo gene mutations that modify their antigen receptors – the molecules they produce to bind to foreign targets. The light zone selects the B cells that bind most tightly and specifically to the target.
The mutations introduced in the dark zone are important because they produce a wide range of B cells that have the best chance of binding tightly to an antigen. The tightest binders are selected because they can fight the infection most efficiently. B cells that don’t bind to the antigen either die or undergo further rounds of mutation. “It’s like Darwinian evolution on a cellular level,” explain Dr Andrew Franklin and Robin Graf, who are coauthors of the study.
Without FOXO1 both mutation and selection are far less efficient. A lack of FOXO1 also affects a process called class switching to the point where the process is almost eliminated. Class switching is needed to produce antibodies with diverse functional classes. These classes have different roles in the immune system such as working in certain parts of the body or fighting particular pathogen types.
The MDC researchers published their work in the journal Immunity where it appeared alongside a paper from a US university that showed similar results. The journal celebrated the important new insights from the two research groups with an artist’s impression of B cell maturation on the journal cover. The cover shows B cells as Alice in Wonderland wondering whether to go down the rabbit hole into the dark zone of the germinal centre. FOXO1 is depicted as a lamp, lighting the way into the dark zone.
In the future, the researchers will focus on how FOXO1 is involved in the early development of lymphomas, since this study has made it clear that the transcription factor plays an important role in the normal germinal centre reaction. It is also known that lymphomas derived from the germinal centre often carry activating mutations of FOXO1.
Sandrine Sander, Van Trung Chu, Tomoharu Yasuda, Andrew Franklin, Robin Graf, Dinis Pedro Calado, Shuang Li, Koshi Imami, Matthias Selbach, Michela Di Virgilio, Lars Bullinger, Klaus Rajewsky (2015): „PI3 Kinase and FOXO1 Transcription Factor Activity Differentially Control B Cells in the Germinal Center Light and Dark Zones.“ Immunity 43, 1075 – 86. doi:10.1016/j.immuni.2015.10.021