Creating protein maps of tumors
To understand what makes malignant cells so dangerous, but also potentially vulnerable, it is not just enough to look at their genes. “It is rather the proteins that are crucial for the functions of all cells,” says Dr. Fabian Coscia, who has headed the Spatial Proteomics Lab at the Berlin-based Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC) since June 2021.
Therefore, during his time as a postdoc in Professor Matthias Mann’s group at the University of Copenhagen’s Novo Nordisk Foundation Center for Protein Research (CPR), Coscia and his colleague Dr. Andreas Mund developed a method that uses artificial intelligence to analyze cancer cell proteomes – the entirety of the proteins produced by the cells. The research teams are now presenting the method, called Deep Visual Proteomics (DVP), in the journal “Nature Biotechnology”.
Peering deeper into tumor tissue
“When something goes wrong in our cells and we get sick, we can be certain that proteins are involved in many different ways,” explains Mann, co-founder of the still relatively young field of proteomics, who not only conducts research in Copenhagen, but also heads the Proteomics and Signal Transduction Department at the Max Planck Institute of Biochemistry in Martinsried near Munich. “So mapping the protein landscape can help us establish why a tumor developed in a particular patient, what the vulnerabilities of that tumor are, and which treatment strategy might be the most useful.”
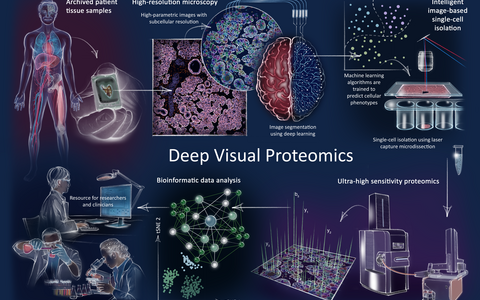
Graphic explaining Deep Visual Proteomics.
Deep Visual Proteomics enables scientists to map the proteins of cancer cells in greater detail than ever before. The technique essentially consists of four steps. First, a high-resolution microscope produces a precise image of the tissue sample. With the help of artificial intelligence, the individual cells in the sample are then classified into different groups on the basis of their visual properties, which are so small that the human eye can barely detect them. Next, an automated laser beam isolates the cells from the tissue, one group at a time, and combines them into a new sample. In the final step, a mass spectrometer determines the exact composition of the proteins in the different samples.
One tool for all cancers
Before proteome analysis, the highly sensitive mass spectrometers must be calibrated.
“By combining state-of-the-art microscopic and mass spectrometric techniques with advances in artificial intelligence, we have gained unprecedented insight into the disease process of cancer,” says Coscia. “We now need fewer than a hundred cells to simultaneously map the thousands of proteins in them.” The method also provides a completely unbiased picture of the processes at work in different cancer cells and may be able to identify still unknown proteins that are involved, for example, in the spread of the cells throughout the body and thus in the development of metastases. Deep Visual Proteomics can also better capture interactions between cancer cells and the surrounding tissue.
In their current paper, the researchers limited their analysis to tumor cells from skin and salivary gland cancer patients. “But the technique can, of course, also be used to characterize all other tumor types in more detail,” says Coscia. The scientist now plans to examine other kinds of tumors as part of the project MSTARS (Multimodal Clinical Mass Spectrometry for the Study of Therapy Resistance), whose participants include the MDC, Charité – Universitätsmedizin Berlin, the Max Planck Institute for Molecular Genetics, and the Humboldt-Universität zu Berlin.
Applicable to other diseases too
“One of the good things about proteins is that they are so stable,” says Coscia, adding that the new method allows them to investigate formalin-fixed patient samples that are many years old and compare the results with the patients’ clinical data. “In this way we want to find out why a certain therapy may have worked particularly well in some cases, but unfortunately failed in others,” the researcher explains. His goal, he says, is to identify new targets for personalized, patient-tailored cancer therapies, even for previously treatment-resistant tumors.
Group leader Fabian Coscia checks the samples before they are analyzed.
However, the capabilities of Deep Visual Proteomics do not stop there. The method cannot only be used to better understand cancer, but also to shed more light on other diseases. “It enables you, for instance, to analyze the proteins in neurons to find out exactly what happens in the nerve cells during neurodegenerative diseases such as Alzheimer’s or Parkinson’s,” says Coscia. He is very much looking forward to seeing in the future how the tool that he and his colleagues developed will be used to gain new biomedical insights by researchers across many different disciplines.
Text: Anke Brodmerkel
Further information
- Berlin-based proteomics consortium “MSTARS”
- Fabian Coscia seeks to shed new light on cancer cells
- Research department “Proteomics and Signal Transduction” at the Max Planck Institute of Biochemistry in Munich
Literature
Andreas Mund, Fabian Coscia et al. (2022): „Deep Visual Proteomics defines single cell identity and heterogeneity“. Nature Biotechnology, DOI: 10.1038/s41587-022 – 01302‑5
Pictures to download
- Before proteome analysis, the highly sensitive mass spectrometers must be calibrated. Photo: Felix Petermann, MDC
- Group leader Fabian Coscia checks the samples before they are analyzed. Photo: Felix Petermann, MDC
Contacts
Dr. Fabian Coscia
Head of the Spatial Proteomics Lab
Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC)
+49-(0)30 – 9406-2532
fabian.coscia@mdc-berlin.de
Jana Schlütter
Editor, Communications Department
Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC)
+49-(0)30 – 9406-2121
jana.schluetter@mdc-berlin.de or presse@mdc-berlin.de
- Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC)
-
The Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC) is one of the world’s leading biomedical research institutions. Max Delbrück, a Berlin native, was a Nobel laureate and one of the founders of molecular biology. At the MDC’s locations in Berlin-Buch and Mitte, researchers from some 60 countries analyze the human system – investigating the biological foundations of life from its most elementary building blocks to systems-wide mechanisms. By understanding what regulates or disrupts the dynamic equilibrium in a cell, an organ, or the entire body, we can prevent diseases, diagnose them earlier, and stop their progression with tailored therapies. Patients should benefit as soon as possible from basic research discoveries. The MDC therefore supports spin-off creation and participates in collaborative networks. It works in close partnership with Charité – Universitätsmedizin Berlin in the jointly run Experimental and Clinical Research Center (ECRC), the Berlin Institute of Health (BIH) at Charité, and the German Center for Cardiovascular Research (DZHK). Founded in 1992, the MDC today employs 1,600 people and is funded 90 percent by the German federal government and 10 percent by the State of Berlin.