ERC Proof of Concept Grant for Mina Gouti
Dr. Mina Gouti and her colleagues are developing highly complex organoids from the reprogrammed stem cells of patients with neuromuscular diseases like spinal muscular atrophy (SMA). “The children experience paralysis in the first months of life, and in the end they can’t even breathe,” says Gouti, who heads the Stem Cell Modeling of Development and Disease Lab at the Max Delbrück Center. “Our neuromuscular organoids, we call them NMOs, can help us understand exactly why the motor neurons of SMA patients die and find ways to stop this process. And that’s just one example of many different diseases that can be studied using NMOs.”
But to develop new therapies – and get robust results – her lab needs to make thousands of similar NMOs at the same time. The researchers are facing the same challenges as the entire field: reproducibility and scalability. “For advanced 3D cell culture systems such as ours to reach their full potential, we need to develop automated, reliable, and high-throughput approaches that are also capable of meeting industry requirements,” Gouti says. The European Research Council (ERC) agrees with this assessment and is now funding her efforts to achieve automation with a Proof of Concept (PoC) Grant of €150,000. Gouti is one of 366 researchers from all over Europe who are receiving PoC funding in the 2022 competition, paving the way for them to translate their findings from ERC Consolidator Grant projects into broadly applied solutions.
Standardized care for organoids
Organoids are a bit like babies: They have to be fed and cared for continuously to keep them happy so that they can mature. “At the moment, most of my lab is busy generating these complex organoids manually,” Gouti says. “It’s labor-intensive and expensive. Plus, the results can vary from one ‘caregiver’ to another.” She wants to automate each step of the process and create a more controlled environment.
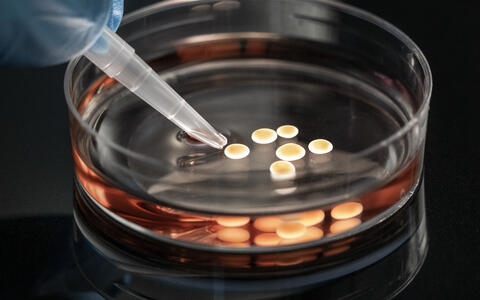
Organoids can live up to two years in the Petri dish.
In order to teach a robot how to nurture organoids, she is teaming up with the Pluripotent Stem Cell Platform of Dr. Sebastian Diecke, which already has an appropriate system in place. Additionally, her lab’s high-throughput imaging system will take pictures of the organoids at various time points, with artificial intelligence assessing their morphology and size. “That is the beauty of working at the Max Delbrück Center,” she says. “It enables this sort of collaboration – one that boosts each other’s research. And if we are successful, we will have robust data and much more time to analyze disease mechanisms.”
High-throughput drug screening, however, poses yet another challenge. Once the organoids are 50 to 100 days old – just mature enough to model neuromuscular diseases – they measure five to six millimeters and are too big to fit into standard 96-well microtiter plates. “We need to miniaturize them,” Gouti says. “So one question is how to keep them small but functional. The other question is: Can we speed up maturation if they don’t need to grow that much?”
Step by step, Gouti and her colleagues are improving the technology, making it available to other labs in Berlin and – hopefully – Big Pharma. “I’m super happy to start this process,” Gouti says. “Our ultimate goal is to establish NMOs as a new preclinical model for drug testing. I’m convinced that they can help to reduce animal studies. And even more importantly, many patients with rare neuromuscular diseases are desperately waiting for therapies.”
Further information
- Mina Gouti awarded ERC Consolidator Grant
- The tissue engineer – a profile of Mina Gouti
- ERC press release about the Proof of Concept Grants
- Einstein Center 3R aims to advance animal-free research
Downloads
-
Portrait of Mina Gouti
Photo: Pablo Castagnola, Max Delbrück Center -
Neuromuscular organoids in the petri dish
Foto: Pablo Castagnola, Max Delbrück Center
Contacts
Dr. Mina Gouti
Head of the Stem Cell Modeling of Development and Disease Lab
Max Delbrück Center
mina.gouti@mdc-berlin.de
Jana Schlütter
Editor, Communications Department
Max Delbrück Center
+49-(0)30 – 9406-2121
jana.schluetter@mdc-berlin.de or presse@mdc-berlin.de
- Max Delbrück Center
-
The Max Delbrück Center for Molecular Medicine in the Helmholtz Association (Max Delbrück Center) is one of the world’s leading biomedical research institutions. Max Delbrück, a Berlin native, was a Nobel laureate and one of the founders of molecular biology. At the locations in Berlin-Buch and Mitte, researchers from some 70 countries study human biology – investigating the foundations of life from its most elementary building blocks to systems-wide mechanisms. By understanding what regulates or disrupts the dynamic equilibrium of a cell, an organ, or the entire body, we can prevent diseases, diagnose them earlier, and stop their progression with tailored therapies. Patients should be able to benefit as soon as possible from basic research discoveries. This is why the Max Delbrück Center supports spin-off creation and participates in collaborative networks. It works in close partnership with Charité – Universitätsmedizin Berlin in the jointly-run Experimental and Clinical Research Center (ECRC), the Berlin Institute of Health (BIH) at Charité, and the German Center for Cardiovascular Research (DZHK). Founded in 1992, the Max Delbrück Center today employs 1,800 people and is 90 percent funded by the German federal government and 10 percent by the State of Berlin.