Muscle cell secrets
Usually, each cell has exactly one nucleus. But the cells of our skeletal muscles are different: These long, fibrous cells have a comparatively large cytoplasm that contains hundreds of nuclei. But up to now, we have known very little about the extent to which the nuclei of a single muscle fiber differ from each other in terms of their gene activity, and what effect this has on the function of the muscle.
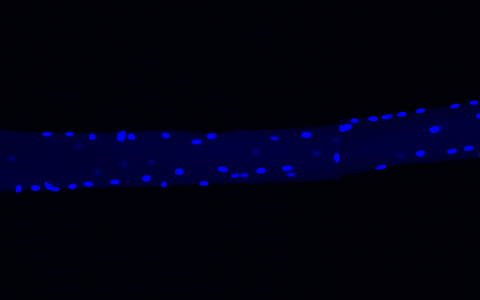
In this single muscle fiber, a multitude of nuclei can be clearly seen. The researchers used DAPI for staining, it stains the DNA in the nuclei blue.
A team led by Professor Carmen Birchmeier, head of the research group on Developmental Biology / Signal Transduction at the Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC), has now unlocked some of the secrets contained in these muscle cell nuclei. As the researchers report in the journal Nature Communications, the team investigated the gene expression of cell nuclei using a still quite novel technique called single-nucleus RNA sequencing – and in the process, they came across an unexpectedly high variety of genetic activity.
Muscle fibers resemble entire tissues
“Due to the heterogeneity of its nuclei, a single muscle cell can act almost like a tissue, which consists of a variety of very different cell types,” explains Dr. Minchul Kim, a postdoctoral researcher in Birchmeier’s team and one of the two lead authors of the study. “This enables the cell to fulfill its numerous tasks, like communicating with neurons or producing certain muscle proteins.”
It is essential that computational labs are part of the process early on as analysis is as important as data generation.
Kim undertook the majority of the experimental work in the study, and his data was also evaluated at the MDC. The bioinformatics analyses were performed by Dr. Altuna Akalin, head of the Bioinformatics and Omics Data Science Platform at the MDC’s Berlin Institute of Medical Systems Biology (BIMSB), and Dr. Vedran Franke, a postdoctoral fellow in Akalin’s team and the study’s co-lead author. “It was only thanks to the constant dialogue between the experiment-based and theory-based teams that we were we able to arrive at our results, which offer important insight for research into muscle diseases,” emphasizes Birchmeier. “New techniques in molecular biology such as single cell sequencing create large amounts of data. It is essential that computational labs are part of the process early on as analysis is as important as data generation,” adds Akalin.
Injured muscles contain activated growth-promoting genes
The researchers began by studying the gene expression of several thousand nuclei from ordinary muscle fibers of mice, as well as nuclei from muscle fibers that were regenerating after an injury. The team genetically labeled the nuclei and isolated them from the cells. “We wanted to find out whether a difference in gene activity could be observed between the resting and the growing muscle,” says Birchmeier.
And they did indeed find such differences. For example, the researchers observed that the regenerating muscle contained more active genes responsible for triggering muscle growth. “What really astonished us, however, was the fact that, in both muscle fiber types, we found a huge variety of different types of nuclei, each with different patterns of gene activity,” explains Birchmeier.
Stumbling across unknown nuclei types
Before the study, it was already known that different genes are active in nuclei located in the vicinity of a site of neuronal innervation than in the other nuclei. “However, we have now discovered many new types of specialized nuclei, all of which have very specific gene expression patterns,” says Kim. Some of these nuclei are located in clusters close to other cells adjacent to the muscle fiber: for example, cells of the tendon or perimysium – a connective tissue sheath that surrounds a bundle of muscle fibers.
We have come across hundreds of genes in previously unknown small groups of nuclei in the muscle fiber that appear to be activated.
“Other specialized nuclei seem to control local metabolism or protein synthesis and are distributed throughout the muscle fiber,” Kim explains. However, it is not yet clear what exactly the active genes in the nuclei do: “We have come across hundreds of genes in previously unknown small groups of nuclei in the muscle fiber that appear to be activated,” reports Birchmeier.
Muscle dystrophy seemingly causes many nuclei types to be lost
In a next step, the team studied the muscle fiber nuclei of mice with Duchenne muscular dystrophy. This disease is the most common form of hereditary muscular dystrophy (muscle wasting) in humans. It is caused by a mutation on the X chromosome, which is why it mainly affects boys. Patients with this disease lack the protein dystrophin, which stabilizes the muscle fibers. This results in the cells gradually dying off.
“In this mouse model, we observed the loss of many types of cell nuclei in the muscle fibers,” reports Birchmeier. Other types were no longer organized into clusters, as the team had previously observed, but scattered throughout the cell. “I couldn’t believe this when I first saw it,” she recounts. “I asked my team to repeat the single-nucleus sequencing immediately before we investigated the finding any further.” But the results remained the same.
The mouse nuclei resemble those of human patients
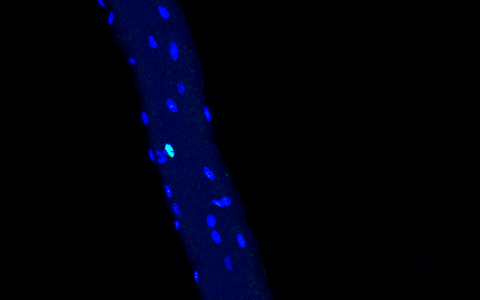
In this part of a muscle fiber, Rian was stained as well. Rian is a long, non-coding RNA (lncRNA) that is highly expressed in a cluster. This indicates that the nuclei have a function in metabolism of the cell.
“We also found some disease-specific nuclear subtypes,” reports Birchmeier. Some of these are nuclei that only transcribe genes to a small extent and are in the process of dying off. Others are nuclei that contain genes that actively repair damaged myofibers. “Interestingly, we also observed this increase in gene activity in muscle biopsies of patients with muscle diseases provided by Professor Simone Spuler’s Myology Lab at the MDC,” says Birchmeier. “It seems this is how the muscle tries to counteract the disease-related damage.”
“With our study, we are presenting a powerful method for investigating pathological mechanisms in the muscle and for testing the success of new therapeutic approaches,” concludes Birchmeier. As muscular malfunction is also observed in a variety of other diseases, such as diabetes and age- or cancer-related muscle atrophy, the approach can be used to better research these changes too. “We are already planning further studies with other disease models,” Kim confirms.
Text: Anke Brodmerkel
Literature
Minchul Kim, Vedran Franke et al. (2020): “Single-nucleus transcriptomics reveals functional compartmentalization in syncytial skeletal muscle cells”. In: Nature Communications, DOI: 10.1038/s41467-020 – 20064‑9.
The Max Delbrück Center for Molecular Medicine
The Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC) was founded in Berlin in 1992. It is named for the German-American physicist Max Delbrück, who was awarded the 1969 Nobel Prize in Physiology and Medicine. The MDC’s mission is to study molecular mechanisms in order to understand the origins of disease and thus be able to diagnose, prevent and fight it better and more effectively. In these efforts the MDC cooperates with the Charité — Universitätsmedizin Berlin and the Berlin Institute of Health (BIH ) as well as with national partners such as the German Center for Cardiovascular Research and numerous international research institutions. More than 1,600 staff and guests from nearly 60 countries work at the MDC, just under 1,300 of them in scientific research. The MDC is funded by the German Federal Ministry of Education and Research (90 percent) and the State of Berlin (10 percent), and is a member of the Helmholtz Association of German Research Centers. www.mdc-berlin.de