Starting up with highly promising immunotherapies
The CARTemis Therapeutics GmbH team (from left to right): Armin Rehm, Anthea Wirges, Uta Höpken and Mario Bunse
Three is the magic number: Working with their labs at the Max Delbrück Center, Dr. Uta Höpken and Dr. Armin Rehm have developed three cell products for potential CAR T‑cell therapies to treat cancer. Two of them will shortly enter their first human trials, while the third is at an advanced stage in the lab. To get their three candidate therapies to the point of regulatory approval, Höpken and Rehm have now founded CARTemis Therapeutics GmbH, a spin-off from the Max Delbrück Center. They are joined by Dr. Anthea Wirges and Dr. Mario Bunse, both research associates working with Höpken and Rehm. The two labs have played a crucial role in moving the CAR T‑cell candidates through preclinical development. Wirges is the start-up’s CEO.
CAR T‑cell therapies are often the last resort for patients with forms of leukemia, myeloma, or lymphoma that do not respond to conventional treatments. The new technique involves taking immune cells (T cells) from the patient and equipping them with a chimeric antigen receptor (CAR) in the laboratory. The CAR acts like a tiny antenna, scanning the body’s cells for specific features of cancer cells. Once the CAR T cells are introduced back into the patient’s body, they can detect and destroy cancer cells carrying the antigen that fits their new receptors.
Fighting cancer with specially equipped T cells
The newest CAR from the CARTemis portfolio fights B non-Hodgkin’s lymphoma, a type of lymph node cancer. It targets a molecule called CXCR5, which is found on mature lymph node cancer cells and on some helper T cells that encourage tumor growth. This CAR T‑cell therapy is set to enter clinical trials next year at Charité – Universitätsmedizin Berlin. The German Federal Ministry of Education and Research (BMBF) is providing €4.6 million in funding. The second CAR targets BCMA, a protein found on the transformed plasma cells in multiple myeloma. A phase 1/2a trial is likely to begin at the end of the year at the National Center for Tumor Diseases in Dresden. The BMBF is providing €1.3 million in funding.
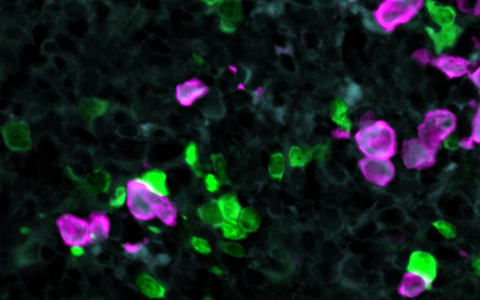
CAR‑T cells in green attack tumor cells (lymphoma cells) in purple.
The third cell product in the pipeline amplifies the effect of CAR T‑cell therapies. It is a lab-produced microRNA that downregulates the protein EBAG9 in CAR T cells. This protein acts like a brake, limiting the release of cell toxins that the CAR T cells use to kill tumor cells. Deactivating EBAG9 has two crucial benefits: First, even a small amount of antigen on a tumor cell will trigger a maximum reaction from the CAR T cells. Second, fewer CAR T cells are needed for therapeutic success overall. The researchers are currently developing an optimal vector system that will allow them to start a clinical trial.
“We’ve really put our heart and soul into these cell products,” says Höpken. “We want to do everything we can to get them approved as cancer therapies and make them available to as many patients as possible.” This means they had no choice but to found the spin-off – public funding would only have taken them to the earliest stages of clinical trials. The Max Delbrück Center supported the group on its way to start-up status through the SPOT program, a funding scheme for teams working on commercially promising products. The Helmholtz Association also provided financing from its spin-off fund.
For CEO Wirges, it’s now time to win over investors. The start-up needs venture capital so it can acquire the licenses from the Max Delbrück Center, prepare the phase 2 trials for their CARs, and continue refining their CAR amplifier. It’s all very new territory for the CARTemis team, but it’s exciting: “We’re thrilled that our research will soon be helping patients,” says Wirges.
Text: Jana Ehrhardt-Joswig