€4.6 million for a novel CAR T‑cell therapy
Joint press release by the Max Delbrück Center and Charité – Universitätsmedizin Berlin
CAR T‑cell therapies are often the last resort for patients with certain types of leukemia and lymphoma who do not respond to standard treatments. This technique involves taking immune cells (T cells) from the patient and equipping them with a chimeric antigen receptor (CAR) in the laboratory. The CAR acts like a tiny antenna, patrolling the body’s cells for specific features on the cancer cells. Once the CAR T cells are re-introduced back into the patient’s body, they begin to detect and destroy cancer cells with the antigen that fits their new receptors.
The research labs of Dr. Uta Höpken and Dr. Armin Rehm at the Max Delbrück Center have developed a novel approach to CAR‑T cell therapy for B non-Hodgkin’s lymphomas, a type of lymph node cancer that originates from B lymphocytes. Under the direction of Professor Ulrich Keller and Professor Lars Bullinger of Charité – Universitätsmedizin Berlin, the new immunotherapy will be tested in humans for the first time in a phase I/IIa trial. The German Federal Ministry of Education and Research (BMBF) is funding the joint project of Charité and the Max Delbrück Center to the tune of €4.6 million. In doing so, it aims to bring forward therapies to tackle currently untreatable or difficult to treat diseases.
CXCR5: An easy target for CARs
The CARs approved so far to treat leukemias and lymphomas usually dock to the antigen CD19, a surface molecule on B cells that can undergo malignant transformation and become cancerous. Höpken and Rehm have identified a molecule called CXCR5 that is likely an even better target for a CAR T‑cell therapy against lymphomas. Unlike CD19, which may be present on cancer cells in varying amounts from patient to patient and can even be lost, CXCR5 is found uniformly on all mature lymphoma cells. Another advantage is that it is found not only on tumor B cells, but also on certain T helper cells that promote tumor growth. “These properties make CXCR5 a unique target for CAR T‑cell therapies,” says Höpken. In mouse models, the researchers were able to show that the CAR T cells are particularly good at finding CXCR5 and eliminating the tumor cells. Clinical trials will determine whether the new immunotherapy is safe and effective in humans. Together with his team, Professor Ulrich Keller, Director of Charité’s Department of Hematology, Oncology and Tumor Immunology at Campus Benjamin Franklin, is preparing initial studies with a few selected patients: “We are confident that this phase I study will demonstrate the safety of the new CXCR5-CAR‑T cell therapy and also find initial indications of its efficacy. What makes this approach promising and highly innovative is that it therapeutically addresses both the tumor cell and its supporting microenvironment.”
The team will begin recruiting up to 24 subjects as soon as they receive approval from the Berlin State Office for Health and Social Affairs (LAGeSo) for the cell product manufacturing and from the Paul-Ehrlich-Institut (PEI), the Federal Institute for Vaccines and Biomedicines, for the clinical trial. The scientists expect these first steps will be completed by early 2024. Initially, the trial will include only patients in whom the standard therapy has not worked.
Text: Jana Ehrhardt-Joswig
- Clinical trials
-
Before new drugs are approved for regular clinical use, they are tested for safety and efficacy in a standardized procedure. In a phase I clinical trial, a therapeutic agent is used in humans for the first time – after extensive preliminary testing – in order to obtain preliminary data on tolerability and safety as well as further effects on the organism. The number of participants is small. If no serious side effects occur and there are initial indications of possible efficacy, a phase II clinical trial follows. Here, tolerability and side effects are determined in a somewhat larger number of patients and the dosage is optimized with regard to possible efficacy. Only in phase III trials, which often last for years and include a large number of participants, can proof of the efficacy of the new substance be provided. For this purpose, the new substance is compared with other available and already approved drugs. Only phase III clinical trials provide the data needed for regulatory approval.
Further information
Download
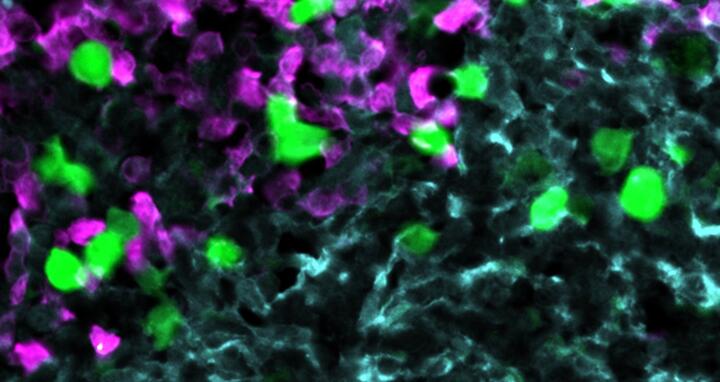
Anti-CXCR5 CAR T cells (green) attack lymphoma cells (red) in the stromal cell network that underpins the B‑cell follicle (light blue). Photo: Höpken Lab / Rehm Lab, Max Delbrück Center
Contacts
Dr. Uta Höpken
Head of the Lab “Microenvironmental Regulation and Autoimmunity in Cancer”
Max Delbrück Center
+49 (0)30 9406 3330
uhoepken@mdc-berlin.de
Dr. Armin Rehm
Head of the Lab “Translational Tumorimmunology”
Max Delbrück Center
+49 (0)30 9406 3817
arehm@mdc-berlin.de
Jana Schlütter
Editor, Communications
Max Delbrück Center
+49 (0)30 9406 2121
jana.schluetter@mdc-berlin.de or presse@mdc-berlin.de
- Max Delbrück Center
-
The Max Delbrück Center for Molecular Medicine in the Helmholtz Association (Max Delbrück Center) is one of the world’s leading biomedical research institutions. Max Delbrück, a Berlin native, was a Nobel laureate and one of the founders of molecular biology. At the Center’s locations in Berlin-Buch and Mitte, researchers from some 70 countries analyze the human system – investigating the biological foundations of life from its most elementary building blocks to systems-wide mechanisms. By understanding what regulates or disrupts the dynamic equilibrium in a cell, an organ, or the entire body, we can prevent diseases, diagnose them earlier, and stop their progression with tailored therapies. Patients should benefit as soon as possible from basic research discoveries. The Max Delbrück Center therefore supports spin-off creation and participates in collaborative networks. It works in close partnership with Charité – Universitätsmedizin Berlin in the jointly run Experimental and Clinical Research Center (ECRC), as well as with the Berlin Institute of Health (BIH) at Charité and the German Center for Cardiovascular Research (DZHK). Founded in 1992, the Max Delbrück Center today employs 1,800 people and is funded 90 percent by the German federal government and 10 percent by the State of Berlin.
- Charité – Universitätsmedizin Berlin
-
Charité – Universitätsmedizin Berlin is one of the largest university hospitals in Europe, boasting 3,099 beds and approximately 100 departments and institutes spread across 4 separate campuses. At Charité, the areas of research, teaching and medical care are closely interlinked. With a total of 20,921 members of staff employed across its group of companies (17,615 of which at Charité), the organization is one of the largest employers in Berlin. 5,047 of its employees work in the field of nursing, with a further 4,988 in research and medical care. Last year, Charité treated 123,793 in- and day case patients, in addition to 682,731 outpatients. In 2021, Charité recorded a turnover of approximately € 2.3 billion (including external funding and investment grants) and set a new record by securing more than € 215.8 million in external funding. Charité’s Medical Faculty is one of the largest in Germany, educating and training more than 9,000 students across the subjects of medicine, dentistry, health sciences and nursing. Charité also offers 730 training positions across 11 different health care professions, in addition to 111 training positions in a further 8 professions. Within the field of academic medicine, Charité’s priorities are highlighted by its main areas of research focus: infection; inflammation and immunity including COVID-19 research; cardiovascular research and metabolism; neuroscience; oncology; regenerative therapies; and rare diseases and genetics. Examples of the work conducted by Charité researchers include involvement in 28 DFG Collaborative Research Centers (of which seven are led by Charité), three Clusters of Excellence (of which one is led by Charité), 10 Emmy Noether Independent Junior Research Groups, 14 European Research Council grants and 8 European collaborative projects (coordinated by Charité).