Targeting a chronic pain gateway could bring relief
Something like a quarter of the world’s population suffers from chronic pain at some point in their lives. As opposed to acute pain – for example, the feeling after hitting your finger with a hammer — chronic pain may not even have a clear cause, and it can linger for years or lifetimes. The burden of chronic pain includes damage to mental and physical health, lower productivity and drug addiction.
A new study led by scientists at the Weizmann Institute of Science (WIS) in Rehovot (Israel) suggests an original approach to treating this affliction, by targeting a key gateway leading to the activation of genes in the peripheral nerve cells that play a role in many forms of chronic pain. The findings of this study were published today in Science.
Pain starts in the sensory neurons – those that pass information from the skin to the central nervous system. Damage to these neurons, chronic injury or disease can cause the neurons to “short circuit,” sending continuous pain messages. Professor Mike Fainzilber of the Biomolecular Sciences Department at WIS investigates molecules that regulate the biomolecular messaging activities taking place within these nerve cells. These molecules – importins – are found in every cell, acting as conduits between the cell nucleus and its cytoplasm, shuttling molecules in and out of the nucleus and thus controlling access to the genes. This role takes on special significance in the peripheral nerve cells, with their long, thin bodies in which molecular messages can take hours to get from nerve endings to cell nuclei. Some of the importins Fainzilber and his team have identified, for example, relay messages about injury to the body of the nerve cell, initiating repair mechanisms.
For this purpose we genetically modified mice so that in each line one of these importins was missing.
A protein that controls a pain pathway
Already for years Professor Michael Bader as head of the lab “Molecular Biology of Peptide Hormones” at the Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC) investigated the importance of importins in a long-term project together with the Institute of Biology at the University of Lübeck. “For this purpose we genetically modified mice so that in each line one of these importins was missing,” Bader explains. To ask which importin is involved in chronic neuropathic pain, the researchers, led by Dr. Letizia Marvaldi in Fainzilber’s group, first set out to screen several importin-mutant mouse lines from Bader’s lab. The research was supported by the European Research Council.
Behavioral screens on these different lines revealed importin alpha‑3 as the only importin implicated in controlling pain pathways. The team then sought to identify the gene expression pattern associated with long-lasting pain in peripheral nerve cells, and see how it tied into importin alpha‑3 activity. Analyses of differences in the expression patterns between normal neurons and neurons lacking importin alpha‑3 directed Marvaldi’s attention to c‑Fos, a protein that importin alpha‑3 brings into the nucleus. c‑Fos is a transcription factor – a molecule that raises or lowers the expression of numerous genes. Further experiments in mice showed that c‑Fos accumulates in the nucleus in peripheral nerve cells of mice suffering from chronic pain.
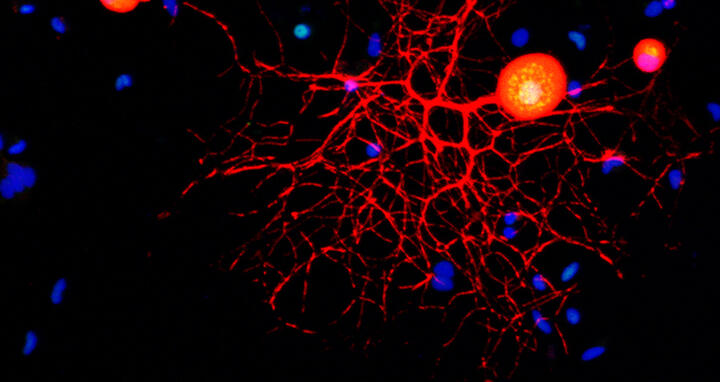
Confocal micrograph of a peripheral sensory neuron in culture. Marker stains and antibodies are used to identify neurons (red), c‑Fos protein (green) and nuclei (blue). Note the nuclear localization of c‑Fos.
They then used specialized viruses as tools to reduce or disable importin alpha‑3 or c‑Fos in mouse peripheral nerve cells. These mice had much reduced responses to chronic pain situations than those of regular mice. Further research showed that importin alpha‑3 is critical in late and chronic pain. c‑Fos is also involved in earlier pain responses, but it seems to enter the nucleus by other means at those earlier stages. This suggests that blocking importin alpha‑3 activity might be especially well-suited to preventing lasting, chronic pain.
A potential drug target
The research team then took their findings to the next level, asking how easily they can be translated to clinical application. They took advantage of a specialized database, the Connectivity Map (CMap) from the Broad Institute in Massachusetts, which reveals connections between drugs and gene expression patterns. This database enabled them to identify around 30 existing drugs that might target the importin alpha‑3 — c‑Fos pathway. Almost two thirds of compounds they identified were not previously known to be associated with pain relief. The team chose two – one a cardiotonic drug and the other an antibiotic – and tested them again in mice. Indeed, injection with these compounds provides relief of neuropathic pain symptoms in mice.
“The compounds we identified in this database search are a kind of fast track – proof that drugs already approved for other uses in patients can probably be repurposed to treat chronic pain,” says Marvaldi. “Clinical trials could be conducted in the near future, as these compounds have already been shown to be safe in humans.”
“We are now in a position to conduct screens for new and better drug molecules that can precisely target this chain of events in the sensory neurons,” says Fainzilber. “Such targeted molecules might have fewer side effects and be less addictive than current treatments, and they could provide new options for reducing the burden of chronic pain.” Also participating in this research were Dr. Nicolas Panayotis, Dr. Stefanie Alber, Dr. Shachar Y. Dagan, Dr. Nataliya Okladnikov, Dr. Indrek Koppel, Agostina Di Pizio, Didi-Andreas Song, Yarden Tzur, Dr. Marco Terenzio, Dr. Ida Rishal and Dr. Dalia Gordon, all of the Weizmann Institute of Sciences Biomolecular Sciences Department; Dr. Franziska Rother of the MDC and the University of Lübeck; and Professor Enno Hartmann of the University of Lübeck.
Professor Michael Fainzilber’s research is supported by the Moross Integrated Cancer Center; the David Barton Center for Research on the Chemistry of Life; the Nella and Leon Benoziyo Center for Neurological Diseases; the Laraine and Alan A. Fischer Laboratory for Biological Mass Spectrometry; the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation; the Rising Tide Foundation; Lawrence Feis; the estate of Florence and Charles Cuevas; the estate of Lilly Fulop; the estate of Lola Asseof; and the European Research Council. Professor Fainzilber is the incumbent of the Chaya Professorial Chair in Molecular Neuroscience.
The original version of this press release was distributed by WIS.
Further information
Literature
Letizia Marvaldi et al. (2020): “Importin α3 regulates chronic pain pathways in peripheral sensory neurons”, Science, DOI: 10.1126/science.aaz5875
Downloads
Confocal micrograph of a peripheral sensory neuron in culture. Marker stains and antibodies are used to identify neurons (red), c‑Fos protein (green) and nuclei (blue). Note the nuclear localization of c‑Fos. Foto: Fainzilber/WIS
Press Contacts
Professor Michael Bader
Head of the lab “Molecular Biology of Peptide Hormones”
Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC)
+49 (0)30 9406 2193
mbader@mdc-berlin.de
Christina Anders
Editor, Communications Department
Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC)
+49 (0)30 9406 2118
christina.anders@mdc-berlin.de or presse@mdc-berlin.de
- The Max Delbrück Center for Molecular Medicine (MDC)
-
The Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC) is one of the world’s leading biomedical research institutions. Max Delbrück, a Berlin native, was a Nobel laureate and one of the founders of molecular biology. At the MDC’s locations in Berlin-Buch and Mitte, researchers from some 60 countries analyze the human system – investigating the biological foundations of life from its most elementary building blocks to systems-wide mechanisms. By understanding what regulates or disrupts the dynamic equilibrium in a cell, an organ, or the entire body, we can prevent diseases, diagnose them earlier, and stop their progression with tailored therapies. Patients should benefit as soon as possible from basic research discoveries. The MDC therefore supports spin-off creation and participates in collaborative networks. It works in close partnership with Charité – Universitätsmedizin Berlin in the jointly run Experimental and Clinical Research Center (ECRC), the Berlin Institute of Health (BIH) at Charité, and the German Center for Cardiovascular Research (DZHK). Founded in 1992, the MDC today employs 1,600 people and is funded 90 percent by the German federal government and 10 percent by the State of Berlin.