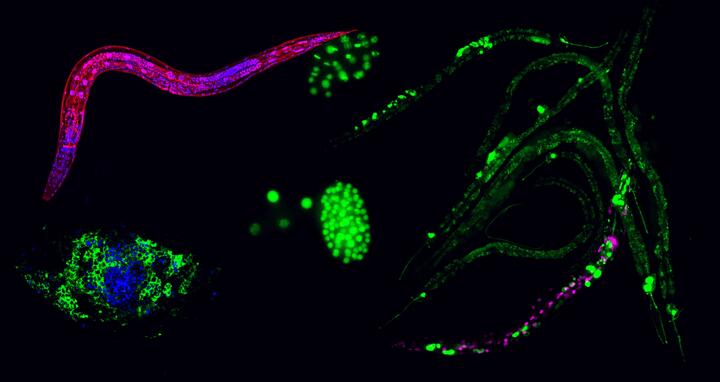
Tiny worms are helping reprogram human cells
What if you walked through the soles of your favorite shoes? You’d ask your shoemaker to replace them with a new pair. But fixing a diseased organ – such as dystrophic muscle or an Alzheimer’s‑riddled brain – by swapping broken cells with a bunch of fresh ones is not that easy. But regenerative medicine wants to achieve just that.
The cells of the body can be reprogrammed to any kind of “fresh” cell. But the procedure is complicated, error-prone and not very efficient. Researchers at the Max Delbrück Center for Molecular Medicine (MDC) in Berlin have now found a way to improve the procedure for gaining of these cells. They turned off genes that protect the identity of a cell and efficiently turned skin cells into stem cells or neurons through a method known as “cellular reprogramming.” They published their results in the current issue of the journal Developmental Cell.
Like a worm in a haystack
Reprogramming cells in the lab ideally involves changing the activity of very few key genes. But the process is not very efficient since there are plenty of “cellular barriers”: molecular programs that prevent one type of body cell from turning into another type.
Searching for cellular barriers, Baris Tursun’s research group at the MDC’s Berlin Institute for Medical Systems Biology (BIMSB) screened millions of tiny roundworms (Caenorhabditis elegans).
Ena Kolundzic, a PhD student working in Tursun’s lab, used a technique called RNA interference to turn off each of the worm’s 20,000 genes one by one and test when reprogramming of cells in the animals is enhanced. Through this tour de force, she identified 170 genes that block reprogramming in the worm and act as a barrier.
An unexpected candidate
Among the found barriers, the genes coding for the protein complex called FACT (facilitates chromatin transcription) caught the attention of the scientists. Tursun and colleagues revealed that FACT “in fact” safeguards the identity of cells, such that switching it off makes cells more susceptible to the reprogramming procedure and improves efficiency.
This is true not only for worms, but also in human skin cell cultures (fibroblasts). Together with colleagues of the MDC’s Pluripotent Stem Cell Platform, the scientists produced 50 percent more neuronal cells and doubled the number of induced pluripotent stem cells (iPSCs) from the same amount of starting material.
Thinking out of the box
“We used a rather unconventional approach to identify cellular barriers,” says Kolundzic. People usually start out with mammalian cell cultures, she says. “But we started with an in vivo system – the worms – and discovered a stunning degree of evolutionary conservation along the way.”
It will take time until these results will benefit patients, Tursun says. But in his view, the findings are remarkable nonetheless: “The identification of the barriers that prevent reprogramming is one of the most pressing issues in regenerative therapy.”
That’s why further genetic screens using C. elegans may provide even more unpredicted targets, the researcher elaborates, adding: “This would aid future tissue replacement therapy” – the equivalent of swapping worn-out cells for new ones and healing incurable diseases.
Reference
Ena Kolundzic et al. (2018): “FACT Sets a Barrier for Cell Fate Reprogramming in Caenorhabditis elegans and Human Cells,” Developmental Cell 46. doi: 10.1016/j.devcel.2018.07.006