Chaos in lymph nodes promotes cancer
Joint press release by the Berlin Institute of Health at Charité (BIH) and the Max Delbrück Center
Lymph nodes are highly organized command centers of the immune system. Within these structures, immune cells gather information about invading pathogens or abnormal cells and coordinate a precise defense response. This finely tuned collaboration enables the body to fight infections and cancer effectively. Crucial to this success is spatial organization: distinct immune cell types occupy defined zones within the lymph node and carry out specialized tasks. This complex tissue architecture forms the foundation of an effective immune response.
In lymphoma, however, this order begins to unravel. Malignant immune cells can profoundly alter the architecture of the lymph node. In some lymphoma subtypes, spatial organization remains largely intact. In others — particularly aggressive forms — it breaks down completely. Although these structural differences have long been used diagnostically, it has remained unclear why certain lymphomas destroy tissue architecture and how this influences disease progression.
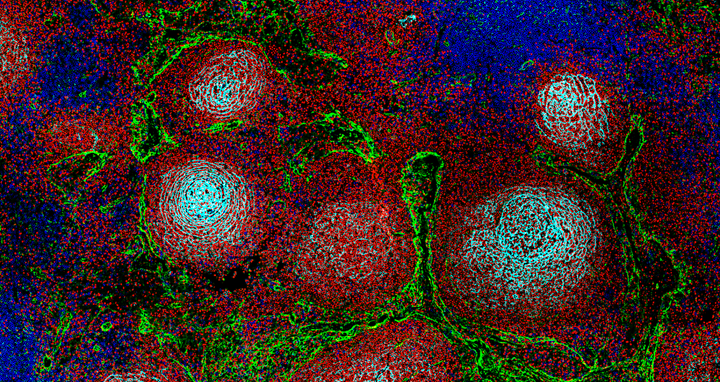
Structure of a lymph node showing the typical division into different territories for immune cells: B cells are shown in red, T cells in blue, lymphatic vessels in yellow, and stromal cells in cyan.
Now, an international research team has solved this puzzle and mapped the architecture of lymph nodes in patients with different lymphoma subtypes in detail. The research was spearheaded by the labs of Professor Simon Haas (Berlin Institute of Health at Charité, BIH, Max Delbrück Center, Queen Mary University of London), Professor Sascha Dietrich (Department of Hematology, Oncology, and Clinical Immunology at the University Hospital Düsseldorf), Professor Judith Zaugg (European Molecular Biology Laboratory, University of Basel), and Dr. Daniel Hübschmann (German Cancer Research Center, DKFZ). The findings, published in “Nature Cancer,” point to new biomarkers and targeted therapeutic strategies.
Out of sync: Conductors of the immune system
Using high-resolution single-cell and spatial technologies, the researchers compared lymph nodes with preserved architecture to those in which tissue organization had collapsed. “These approaches allow us to trace molecular, cellular and spatial changes within the tissue with great precision,” explains Dr. Lea Jopp-Saile, a first author of the paper and scientist in the lab of Simon Haas.
The team found that rare stromal cells – specialized support cells that permeate the lymph nodes like a fine network – are largely responsible for the spatial organization of immune cells in the lymph nodes. In healthy lymph nodes, they act as “conductors” of the immune system, using biochemical messengers to direct the immune cells to defined areas. In particularly aggressive lymphomas, however, this conducting function is profoundly disrupted.
A vicious inflammatory circle
The driving force behind this loss of tissue organization is a self-reinforcing inflammatory cycle. In an attempt to eliminate the tumor, T cells, which are specialized immune cells, release inflammatory signaling molecules. These signals reprogram the stromal cells, altering their communication patterns. As a result, the stromal cells lose their structural role and the lymph node’s spatial organization collapses. The loss of architecture is therefore not merely a passive consequence of tumor growth, but an active process fueled by inflammation within the tumor microenvironment.
Analyses of large patient cohorts further showed that stromal cell reprogramming is linked to a significantly poorer prognosis. “We believe that by actively dismantling the lymph node’s structure, the tumor weakens the very local immune response that is meant to eliminate it, thereby promoting its own growth,” says Dr. Felix Czernilofsky, also a first author of the paper.
“Our results suggest that stabilizing stromal cells or selectively modulating inflammatory signals could represent promising new treatment approaches,” adds Professor Dietrich, Clinical Director and lymphoma specialist at the Clinic for Hematology, Oncology and Rheumatology at Heidelberg University Hospital. “In addition, the mechanisms we identified may help to develop new biomarkers to detect aggressive disease at an earlier stage.”
- About the research collaboration
The study was made possible through international collaboration and close interdisciplinary cooperation across traditional research boundaries. Leading contributors included lymphoma experts Dr Felix Czernilofsky and Professor Sascha Dietrich (University Hospitals of Düsseldorf and Heidelberg), single-cell technology expert Professor Simon Haas (Berlin Institute of Health at Charité and the Max Delbrück Center in Berlin, as well as Queen Mary University of London), and data scientists PD Dr Daniel Hübschmann (German Cancer Research Center, DKFZ, Heidelberg Institute for Stem cell Technology and Experimental Medicine, HI-STEM), Dr Lea Jopp-Saile (DKFZ, HI-STEM), Dr Anna Mathioudaki (European Molecular Biology Laboratory, EMBL, DKFZ), and Professor Judith Zaugg (EMBL, University of Basel).
Further information
- Haas Lab
- Focus area “Single cell approaches for personalized medicine” with BIH and Charité
Literature
Felix Czernilofsky, Lea Jopp-Saile, Anna Mathioudaki et al. (2026): „Reprogramming of stroma-derived chemokine networks drives the loss of tissue organization in nodal B cell lymphoma’“. In Nature Cancer, DOI: 10.1038/s43018-026 – 01136‑z
Photo for Download
Structure of a lymph node showing the typical division into different territories for immune cells: B cells are shown in red, T cells in blue, lymphatic vessels in yellow, and stromal cells in cyan. Photo: Marc-Andrea Bärtsch, Felix Czernilofsky, Med‑V UKHD
Contacts
Prof. Dr. Simon Haas
Berlin Institute of Health at Charité (BIH)
Berlin Institute for Medical System Biology of the Max Delbrück Center (MDC-BIMSB)
Precision Healthcare University Research Institute (PHURI) der Queen Mary University of London
simon.haas@bih-charite.de or simon.haas@mdc-berlin.de
Jana Schlütter
Editor, Communications and Marketing
Max Delbrück Center
+49 30 9406 – 2121
jana.schluetter@mdc-berlin.de or presse@mdc-berlin.de
- Max Delbrück Center
The Max Delbrück Center for Molecular Medicine in the Helmholtz Association lays the foundation for the medicine of tomorrow through our discoveries of today. At locations in Berlin-Buch, Berlin-Mitte, Heidelberg, and Mannheim, interdisciplinary teams investigate the complexity of disease at the systems level – from molecules and cells to organs and entire organisms. Together with academic, clinical, and industry partners, and as part of global networks, we turn biological insights into innovations for early detection, personalized therapies, and disease prevention. Founded in 1992, the Max Delbrück Center is home to a vibrant, international research community of around 1,800 people from over 70 countries. We are 90 percent funded by the German federal government and 10 percent by the state of Berlin.