The dark side of treating cancer with drugs
When the cells in our body are threatened by malignant changes, inherent self-protection programs kick in and often prevent cancer from developing. Two of these tumor-suppression mechanisms are programmed cell death, known as apoptosis, and the acute triggering of a cellular aging-like program, senescence. They inhibit cell division and the growth of tumors. These programs are also activated during chemotherapy and mediate its anti-tumor effect.
Particularly in tumor cells in which apoptosis is no longer functioning properly, the triggering of cellular senescence is an important, desirable therapy effect to inhibit them from continuing to grow. This was already demonstrated some years ago by the team working with Professor Dr Clemens Schmitt, Deputy Director of the Charité – Universitätsmedizin Berlin’s Medical Department with a focus on hematology, oncology and tumor immunology, Director of the Molecular Cancer Research Center, and head of the research group on “Cancer Genetics and Cellular Stress Responses” at the Max Delbrück Center for Molecular Medicine in the Helmholtz Association.
A stem cell program makes the cells especially aggressive
In its new study the research team observed that the tumor cells whose growth has been inhibited undergo massive epigenetic reprograming when they enter the senescent state, causing various cellular programs to be newly coded. Among others, a stem cell program is triggered that is also known as “tumor stemness”. The term describes the particularly threatening ability of cancer cells to promote tumor growth or even to initiate it, for example when metastases develop.
Since the stem cell function is irrevocably linked with cell division, the cancer researchers investigated whether they could functionally visualize the newly-acquired stemness of previously senescent cells by de-activating individual genes that are imperative for maintaining senescence. And, indeed, the previously senescent tumor cells behaved much more aggressively than the same tumor cells that had never entered the senescent state.
An insight into the clever behavior of tumor cells
Experiments on genetically modified mice underpinned the relevance of these cell culture findings. With the help of new single-cell tracking technology the scientists were also able to show that senescent tumor cells can, albeit seldomly, spontaneously re-enter the cell-division cycle. A comparison of human lymphoma samples (a lymph-node cancer) obtained before therapy and later, after recurrence of the disease in the same patient, suggest that after chemotherapy, previously senescent cells promote particularly aggressive tumor growth if the therapy fails.
“These results are very important in clinical terms because they give us insights into the clever behaviour of tumor cells that manage to assert themselves against what are really very effective cancer therapies,” says Professor Schmitt, who treats lymphoma patients at the Charité on a daily basis and also coordinates research activities within the German Cancer Consortium (DKTK). “Luckily, as part of this research work we were also able to present genetic and drug-based strategies that directly attack and neutralize newly-acquired tumor stemness in previously senescent tumor cells.”
Further information:
Maja Milanovic et al (2017): „Senescence-associated reprogramming promotes cancer stemness“. Nature. DOI:10.1038/nature25167
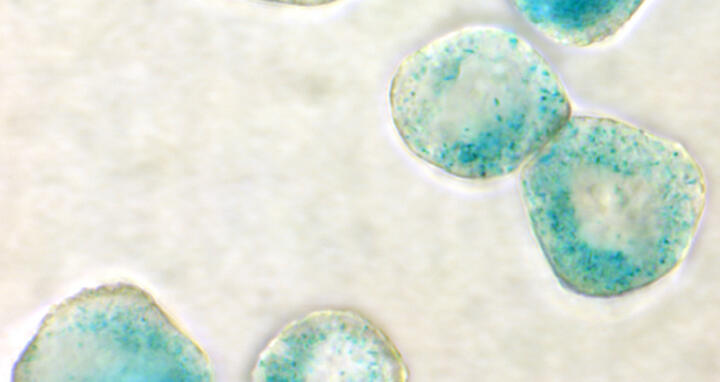
Featured Image: In these lymphoma cells, the therapy has triggered cell ageing (senescence). A senescence-marking blue colour makes the process visible. Image: Schmitt Lab