How bacteria cause tissue changes in the stomach
A joint press release by Charité and the MDC
The infection disrupts complex interactions between different cell types and signals which are responsible for tissue stability.
Affecting about half the world’s population, Helicobacter pylori is one of the most common causes of chronic bacterial infections in humans. H. pylori can lead to inflammation of the stomach (gastritis) and increase the risk of developing stomach cancer. As the protective lining inside the stomach is constantly exposed to stomach acid, it has to regenerate completely every few weeks, while maintaining both its structure and composition despite the high cellular turnover. “Until now, researchers had assumed that a Helicobacter infection causes direct damage to the gastric gland cells in the stomach lining and that gastric pathology upon infection is simply the result of this process,” explains the study’s last author, Professor Michael Sigal. He continues: “In fact, our team has now discovered that the infection disrupts complex interactions between different cell types and signals which are responsible for tissue stability.”
Michael Sigal is Professor for Translational Gastrointestinal Oncology and leads an Independent Research Group at the Department of Hepatology and Gastroenterology at Charité Iniversitätsmedizin Berlin and the Berlin Institute for Medical Systems Biology (BIMSB), which is part of the Max Delbrück Center für Molecular Medicine in the Helmholtz Association (MDC). Led by Sigal and working alongside colleagues from the Max Planck Institute for Infection Biology, the team of researchers used complex mouse models to observe changes occurring inside the gastric glands. Using state-of-the art imaging and single-cell sequencing technologies, the researchers were able to visualize and isolate specific gastric gland cells, which they then examined in detail. In order to minimize the need for animal models, they also developed special organ-like tissue microstructures known as organoids. The researchers used these microscopic stomachs to recreate many of the gastric glands’ characteristics. This strategy enabled them to study the effects of various signals on stem cells which are found inside the gastric glands (and which are capable of differentiating into many different cell types).
Stromal cells inhibit the BMP signaling pathway
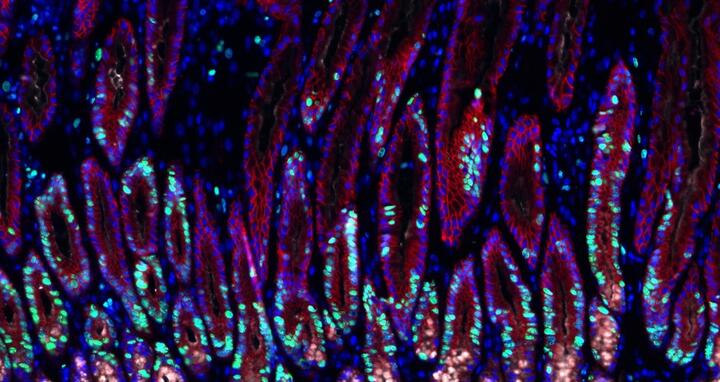
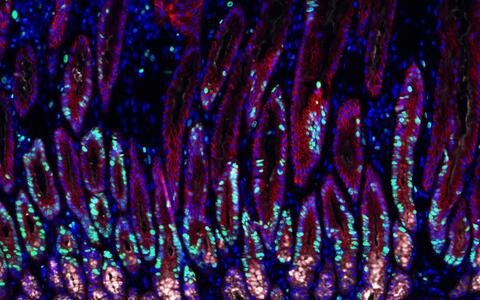
Stomach tissue infected with Helicobacter bacteria. Dividing cells are depicted in green, cell nuclei in blue.
“We discovered that ‘stromal cells’ – a type of cell surrounding the gastric glands – are not only responsible for mechanical stability of the glands, as previously thought. Instead, they produce various signaling molecules which significantly influence the behavior of the gland cells,” explains Prof. Sigal. These substances include ‘bone morphogenetic proteins’ (BMPs), which play an important role in tissue development. The researchers were able to show that stromal cells surrounding the gland base continually inhibit the BMP signaling pathway, thereby stimulating the proliferation of nearby stem cells. In contrast, stromal cells at the gland surface were found to activate the signaling pathway, thereby suppressing cell proliferation. Together, this forms a signaling gradient that guides stem cell turnover and differentiation, and serves as the basis for the gland’s structural stability. Helicobacter infection causes the release of pro-inflammatory substances such as interferon-gamma (IFN‑γ). This inflammatory cytokine interferes with the BMP signaling axis, resulting in less BMP signaling activity and stimulating gland stem cell proliferation. This results in hyperplasia, a precancerous lesion characterized by tissue enlargement.
“Our findings show that an infection-driven inflammatory response has far more pronounced effects than previously thought. In addition to their well-characterized antimicrobial effects, pro-inflammatory substances such as IFN‑γ affect both cell proliferation and tissue stem cell behavior and therefore have a direct impact on tissue homeostasis. In the case of tissue damage, increased cell proliferation can be useful, as it promotes rapid healing. In the case of chronic inflammation associated with a Helicobacter infection, however, it could facilitate the development of precancerous lesions,” summarizes Prof. Sigal. The signaling pathways governing interactions between the immune system and stem cells in the stomach could also prove significant in other organs. As such, they represent a new treatment target – both in cancer prevention and regenerative medicine.
Text: Charité
Further information
Literature
Marta Kapalczynska et al (2022): “BMP feed-forward loop promotes terminal differentiation in gastric glands and is interrupted by H. pylori-driven inflammation”, in: Nature Communications, DOI: 10.1038/s41467-022 – 29176‑w
Image to Download
Stomach tissue infected with Helicobacter bacteria. Dividing cells are depicted in green, cell nuclei in blue. © Charité / Michael Sigal
Contact
Professor Michael Sigal
Department of Hepatology and Gastroenterology
Campus Charité Mitte and Campus Virchow-Klinikum
Charité – Universitätsmedizin Berlin
and Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC)
+49 30 450 614 055
michael.sigal@charite.de
- Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC)
-
The Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC) is one of the world’s leading biomedical research institutions. Max Delbrück, a Berlin native, was a Nobel laureate and one of the founders of molecular biology. At the MDC’s locations in Berlin-Buch and Mitte, researchers from some 60 countries analyze the human system – investigating the biological foundations of life from its most elementary building blocks to systems-wide mechanisms. By understanding what regulates or disrupts the dynamic equilibrium in a cell, an organ, or the entire body, we can prevent diseases, diagnose them earlier, and stop their progression with tailored therapies. Patients should benefit as soon as possible from basic research discoveries. The MDC therefore supports spin-off creation and participates in collaborative networks. It works in close partnership with Charité – Universitätsmedizin Berlin in the jointly run Experimental and Clinical Research Center (ECRC), the Berlin Institute of Health (BIH) at Charité, and the German Center for Cardiovascular Research (DZHK). Founded in 1992, the MDC today employs 1,600 people and is funded 90 percent by the German federal government and 10 percent by the State of Berlin.